Lisa L. Cunningham, Ph.D., Senior Investigator
Research Statement
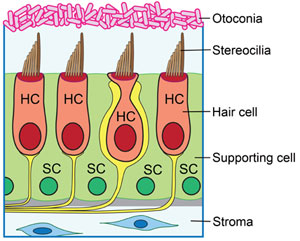
Diagram of cross-section of the utricle showing hair cells and supporting cells.

Localization of cisplatin in the human inner ear using laser ablation inductively-coupled plasma mass spectrometry (ICP-MS). Red and yellow colors indicate higher signal intensity.
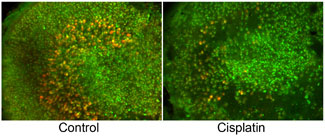
Mouse utricles with hair cells labeled with antibodies against calmodulin (green) and calbindin (red). Left panel is a control utricle showing type I (red) and type II (green) hair cells. Right panel is a utricle treated with cisplatin, which is toxic to hair cells. At this low cisplatin concentration, type I hair cells are killed while type II hair cells survive.
Research in the Section on Sensory Cell Biology (Laboratory of Hearing Biology and Therapeutics) is focused on the mechanosensory hair cells that are the receptor cells of hearing and balance. Hair cells transduce sound energy or head movement into neural input to the brain. Hair cells are sensitive to death from a variety of stresses, including noise trauma, aging, genetic mutations, and exposure to therapeutic drugs with ototoxic side effects. The Section on Sensory Cell Biology conducts basic, translational, and clinical studies on hair cell degeneration and hearing loss in the mature inner ear and develops therapeutic strategies to safeguard hearing.
- Our basic science studies examine the mechanisms that underlie sensory hair cell death and survival.
- Our translational studies use this mechanistic knowledge to guide the rational design of therapies aimed at preventing or reversing hearing loss in humans.
- Our clinical studies test the efficacy of potential protective therapies in humans receiving ototoxic drugs.
Our basic science studies focus on the signal transduction and intercellular communication pathways that are activated in response to hair cell stress and death. We showed that induction of heat shock proteins (HSPs) can protect hair cells against major stresses, including exposure to both classes of ototoxic drugs (i.e., the aminoglycoside antibiotics and cisplatin). Pro-survival induction of HSP expression is relatively low in hair cells and is more robust in surrounding cell types, including the glia-like supporting cells and resident macrophages. These data indicate that hair cells may have a reduced capacity to induce autonomous pro-survival signaling in response to stress, and that non-cell-autonomous signals from supporting cells and macrophages function as critical mediators of pro-survival signaling when hair cells are under stress. Our current basic science studies are designed to examine the intercellular signaling that occurs when hair cells are under stress.
Our translational studies consist of preclinical experiments aimed at developing therapies to preserve hearing in humans exposed to ototoxic drugs. Cisplatin is a widely used drug that is used to treat solid tumors. Cisplatin treatment results in permanent hearing loss for a significant proportion of patients treated with the drug. We have developed a mouse model of cisplatin ototoxicity, and we have used this model to examine the mechanisms underlying cisplatin-induced hearing loss. Our data indicate that cisplatin readily enters the inner ear, but unlike most other organs, the inner ear has very little capacity for eliminating cisplatin. Thus cisplatin remains in the inner ear indefinitely after treatment. Currently, our translational work is aimed at developing therapies to prevent cisplatin uptake into the inner ear.
Our clinical studies are aimed at testing the efficacy of protective therapies in humans taking ototoxic drugs. We are examining the capacity for the cholesterol-lowering drugs known as statins to protect against cisplatin-induced hearing loss in humans. We are conducting a prospective study in patients with head and neck cancer who are undergoing cisplatin-based chemoradiation. The goal of this project is to test the hypothesis that statin use reduces hearing loss in these patients.
Videos
ImageJ Macro Cell Counter Instructional Video
This video demonstrates a method that allows semi-automated quantification of hair cells in the entire utricle by staining adult utricle cultures for Pou4f3, a hair cell-specific transcription factor. The provided utricle images and the ImageJ macro script have been used in the instructional video to demonstrate this method. Users can download these image files and use them to familiarize themselves with this ImageJ macro-based counting method.
- Download Utricle Control (670MB CZI)
- Download Utricle Neomycin (566MB CZI)
- Download Utricle Cisplatin (192MB TIF)
- Download ImageJ Macro Script (3.7KB IJM)
Dissection of Adult Mouse Utricle and Adenovirus-mediated Supporting-cell Infection
This video demonstrates mouse utricle dissection and infection of supporting cells using adenovirus.

Lab staff in 2019. Top row, L-R: Tucker Costain, Melanie Barzik, Benjamin Gersten, Cathy Sung. Bottom row, L-R: Erica Sadler, Katharine Fernandez, Lisa Cunningham, Lizhen (Jenny) Wang.

Cunningham lab group enjoying the DC Wharf, spring 2018.

DC Cherry Blossom Festival 2015. L-R: Shimon Francis, Elyssa Monzack, Lisa Cunningham, Lindsey May, Aaron Rusheen.

Lab staff prior to 2019. Back row, L-R: Shimon Francis, Andrew Breglio. Middle row, L-R: Aaron Rusheen, Lisa Cunningham, Matthew Ryals. Front row, L-R: Katharine Fernandez, Lindsey May.

