Introduction
- NIDCD Overview
- NIDCD Strategic Plan and Priority Setting
- Enhance Scientific Stewardship at the NIDCD
- Shared Databases, Registries, and Metrics on Communication Disorders
- Trans-NIH Efforts Encourage Innovation through Partnerships
- Excel as a Federal Science Agency by Managing for Results
Future Directions in NIDCD Program Areas
Hearing and Balance Research
- Why the NIDCD Supports Hearing and Balance Research
- The Hearing and Balance Program
- Recent Advances in Hearing and Balance Research
- Science Capsule: Balance or Vestibular Disorders in Adults
- Priority Areas in Hearing and Balance Research
Taste and Smell Research
- Why NIDCD Supports Taste and Smell Research
- The Taste and Smell Program
- Recent Advances in Taste and Smell Research
- Science Capsule: How Mosquitoes Target their Human Hosts
- Priority Areas in Taste and Smell Research
Voice, Speech, and Language Research
- Why NIDCD Supports Voice, Speech, and Language Research
- The Voice, Speech, and Language Program
- Recent Advances in Voice, Speech, and Language Research
- Science Capsule: Spasmodic Dysphonia
- Priority Areas in Voice, Speech, and Language
- Summary
- Appendix A: NIDCD Funding History
- Appendix B: The NIDCD 2017-2021 Strategic Plan: The Process
- Appendix C: NIDCD’s Trans-NIH and Trans-Agency Activities
- Appendix D: Glossary and Acronym List
- Appendix E: Bibliography
Welcome from the Director
The National Institute on Deafness and Other Communication Disorders (NIDCD) is pleased to share our new five-year Strategic Plan for 2017-2021. The Plan helps the NIDCD prioritize its research investments by identifying areas of outstanding promise and areas in need of greater funding due to gaps in our knowledge. By prioritizing research investment in these areas, the Institute strives to improve the quality of life for people with communication disorders.
Looking forward, the NIDCD anticipates unprecedented scientific opportunities. We are already using recent advances in science and technology to discover how changes to the molecular, cellular, and systemic pathways can cause communication disorders. The NIDCD hopes to build on these advances by supporting research that will lead to better ways to identify those who are at risk for developing certain communication disorders, with a goal of preventing a disorder from occurring or at least lessening its effects. The NIDCD also continues to support research to develop better treatments for people with communication disorders.
These unprecedented research opportunities are coupled with the challenge of using our best scientific judgment to make difficult choices about which areas of research to pursue. The objectives in this Strategic Plan have been identified through discussions among outside experts in each of the Institute's mission areas, along with input from NIDCD staff members, the National Deafness and Other Communication Disorders (NDCD) Advisory Council, representatives of the research and advocacy communities, and members of the public.
Thank you for your interest in the NIDCD's scientific research. For more information, please visit the NIDCD website at www.nidcd.nih.gov.
Sincerely,
James F. Battey, Jr., M.D., Ph.D.
Director
National Institute on Deafness and Other Communication Disorders
Science Capsule: Advances in Hearing Aid Research
Nearly 15 percent of American adults (37.5 million) aged 18 and over report some trouble hearing, making this one of the most prevalent disabling conditions in the U.S. Hearing loss can be hereditary, or it can result from disease, trauma, medications, or long-term exposure to damaging noise. The condition can vary from a mild but important loss of sensitivity to a total loss of hearing.
Sensorineural hearing loss is caused by a problem in the cochlea or the auditory nerve, which are parts of the ear that help sound impulses reach the brain. Hearing loss affects people of all ages, in all segments of the population, and across all socioeconomic levels. It can interfere with an individual’s physical, cognitive, behavioral, and social functions, and hearing aids are the main form of treatment. However, of adults aged 70 and older with hearing loss who could benefit from wearing hearing aids, fewer than 30 percent have ever used them. Of adults aged 20 to 69 who could benefit from hearing aids, the proportion that has used them is even lower (only about 16 percent).
A hearing aid works by amplifying sound to allow people to hear sounds that would not be audible. In specially equipped movie theaters, auditoriums, lecture halls, places of worship, and other areas, people can use a hearing aid to access “hearing loop” wireless signals that are beamed directly to the aid to bypass background noises. A vast array of hearing aid technology is available to provide additional features, such as the telecoil needed to pick up the hearing loop wireless signal.
Although the development of microelectronic components has enabled new digital hearing aid technology to replace earlier devices based on analog circuits, the underlying damage to the inner ear remains a limitation when the user is confronted by multiple speakers or background noise. Hearing aid users often complain of straining to focus on a single speech sound among competing sources at meetings, banquets, and sporting events. One solution to this problem is to move the hearing aid user closer to the person speaking and farther from the noise sources. Directional microphones offer another approach to do the same thing simply by pointing a device.
NIDCD-supported scientists have studied the remarkable directional hearing of the tiny fly Ormia ochracea, which inspired development of a novel directional microphone to improve hearing aids. Scientists reverse-engineered the physics and biology behind the fly’s abilities to localize sound and provided engineers with strategies to improve directional microphones that are small enough to use in hearing aids and help focus the aid on one sound source at a time.
Capitalizing on the knowledge learned from studying Ormia, another group of NIDCD-supported scientists successfully completed design and testing of a novel microphone based on these design elements. The scientists used silicon microfabrication technology to build the critical sensing elements needed for a functional microphone, characterize its function, and prove it had the capability to provide performance gains over existing designs.
Other NIDCD-supported scientists have continued research and development efforts based on this proof of concept prototype by adapting the microphone design into a form that could be more readily incorporated in a hearing aid. The scientists are the first to use piezoelectric materials, which turn mechanical pressure into electrical signals (voltage) and allow the microphone to operate with very little power. Because hearing aids rely on batteries, minimizing power consumption is a crucial design requirement.
The NIDCD recognizes that the needs of the majority of adults with hearing loss are not being met, and the cost and accessibility of hearing aids are considered part of the barriers to care. In response, the NIDCD is working to fill this need by supporting research or infrastructure that will lead to more accessible and affordable hearing health care for adults. The NIDCD cosponsored a consensus development study with the National Academies of Sciences, Engineering, and Medicine to consider hearing health care from the health care and population health perspectives, including the regulatory environment, access, and affordability. By identifying the research gaps related to effective and affordable hearing health care, devices, and compliance, and by developing novel strategies to overcome these gaps, NIDCD clinical and translational research will endeavor to improve the quality of life for millions of Americans with hearing loss.
Introduction
NIDCD Overview
Approximately 46 million Americans experience some form of communication disorder. Communication disorders make the basic components of communication (sensing, interpreting, and responding to people and things in our environment) challenging. In addition, communication disorders not only compromise physical health, but also affect the emotional, social, recreational, educational, and vocational aspects of life. The effects often ripple outward to affect families and social networks, including those at work and school. The total economic impact of these disorders in regards to quality of life and unfulfilled potential is substantial. Furthermore, the prevalence of communication disorders is expected to increase as the population ages, and as survival rates improve for medically fragile infants and people affected by traumatic injuries and diseases.
In October 1988, Congress established the National Institute on Deafness and Other Communication Disorders (NIDCD) as one of the institutes that compose the National Institutes of Health (NIH), part of the U.S. Department of Health and Human Services. The NIH is the federal government’s focal point for the support of biomedical research and is among the leading biomedical research funding institutions in the world. NIH’s mission is to seek fundamental knowledge about the nature and behavior of living systems and to apply that knowledge to enhance health, lengthen life, and reduce the burdens of illness and disability. NIDCD’s focus within this broad mission is to bring national attention to the disorders and dysfunctions of human communication and to contribute to advances in biomedical and behavioral research that will improve the lives of the millions of people with a communication disorder.
The NIDCD mission is to conduct and support biomedical research, behavioral research, and research training in the normal and disordered processes of hearing, balance, taste, smell, voice, speech, and language.
The Institute conducts and supports research and research training related to disease prevention and health promotion; addresses special biomedical and behavioral problems associated with people who have communication impairments or disorders; supports research evaluating approaches to the identification and treatment of communication disorders and patient outcomes; and supports efforts to create devices that substitute for lost and impaired sensory and communication function.
To accomplish these goals, the NIDCD manages a broad portfolio of both basic and clinical research. The portfolio is organized into three program areas: hearing and balance; taste and smell; and voice, speech, and language. The three program areas seek to answer fundamental scientific questions about normal function and disorders and to identify patient-oriented scientific discoveries for preventing, screening, diagnosing, and treating disorders of human communication. See Appendix A for the NIDCD Funding History.
The NIDCD accomplishes its research mission through three divisions: the Division of Intramural Research (DIR), the Division of Scientific Programs (DSP), and the Division of Extramural Activities (DEA). The DIR conducts research and related support activities in laboratories and clinics housed at the NIH. The DSP and DEA manage complementary aspects of the NIDCD’s Extramural Research Program, a program of research grants, career development awards, individual and institutional research training awards, center grants, and contracts to public and private research institutions and organizations throughout the U.S. and abroad. As a whole, the Institute supported approximately 1,300 research grants, training awards, and research and development contracts in Fiscal Year (FY) 2016. Through research and education, the NIDCD strives to reduce both the direct and indirect economic burden of communication disorders on individuals, families, and society, thereby improving the quality of life for people living with a communication disorder.
NIDCD Strategic Plan and Priority Setting
The NIDCD uses the NIH system of peer review to evaluate research grant applications. The system depends on scientists to submit their best research ideas to drive the spectrum of supported research. The NIH is committed to a transparent, evidence-based process of structured peer review. A panel of scientific experts from outside of the NIH (who work in the same or a related academic field) scrutinize grant applications. To identify research ideas with the highest overall potential impact, the panel evaluates applications for approach, significance, innovation, investigator(s), and quality of the academic environment. This system helps NIH select the most promising ideas to receive federal funding. To learn more about the NIH peer review process, see http://grants.nih.gov/grants/peer/peer.htm. To learn how NIH continuously reviews and updates its peer review process, see http://grants.nih.gov/grants/peer/continuous_review.htm.
The NIDCD values investigator-initiated applications submitted to NIH that help achieve the NIDCD mission. In particular, the Institute encourages investigators to submit applications for research projects that directly address priorities within the NIDCD Strategic Plan (Plan). The NIDCD also uses the Plan to develop targeted Funding Opportunity Announcements (FOAs) to stimulate research applications that address a particular and much-needed area of science.
The NIDCD Strategic Plan helps the Institute (including NIDCD staff and the NDCD Advisory Council) prioritize research investment. The Plan helps identify investigator-initiated research proposals for High Program Priority (HPP) funding so that these projects, if funded, will address a significant research need in the NIDCD portfolio. The NIDCD uses its HPP process to fill scientific gaps in the research portfolio, foster the entry of new investigators, encourage innovative research, and increase the diversity of the scientists who lead a research team, known as Principal Investigators (PIs).
NIDCD staff distribute the Plan to the research community at workshops and scientific conferences to increase awareness of Institute priorities. Additionally, the Plan informs the public about the state of the science and advances in diagnosis and treatment of communication disorders, while creating a vision for the future. To develop the 2017-2021 Plan, the NIDCD convened a series of working group meetings and solicited input from scientific experts, the NDCD Advisory Council, NIDCD staff, and the public. See Appendix B for more details on the Plan process.
Enhance Scientific Stewardship at the NIDCD
Research Training and Career Development at the NIDCD
The number of Americans with communication disorders is expected to rise as the nation’s older population increases and as survival rates improve for a wide range of medical conditions associated with communication disorders. As such, the NIDCD recognizes the importance of research training and career development opportunities to ensure a productive, creative, and innovative cadre of qualified scientists in basic, clinical, and translational research. The NIDCD is continuously adapting its research training and career development efforts to help new scientists establish careers in our mission areas, encourage clinicians to pursue opportunities in translational research, and build shared research resources.
The field of human communication sciences needs interdisciplinary research teams of clinicians and basic scientists to bridge the gap between laboratory research and patient care. Clinicians need a deeper understanding of the latest research discoveries to bring new diagnostic and treatment approaches into the clinic. Basic researchers need a thorough understanding of the needs, challenges, and opportunities faced by clinicians. The NIDCD believes that cross training these scientists could spark new ways to better prevent, detect, and treat communication and chemosensory disorders. Interdisciplinary teams of basic scientists and clinicians—including physicians, surgeons, and audiologists—will then be able to initiate and support new directions for scientific discovery, conduct hypothesis-driven clinical trials, assess new diagnostic tools and interventions, and improve public health and well-being.
Workforce Diversity at the NIDCD
Because human communication disorders cross all social and ethnic groups, the NIDCD recognizes the benefit of a diverse interdisciplinary workforce to tackle the world’s diverse public health needs. In addition, the NIDCD recognizes the underrepresentation of minority scientists in its research and research training activities and diligently works to increase participation of researchers from underrepresented groups. To this end, the NIDCD has made it a priority to increase the number of minorities, individuals with communication disorders, and individuals and groups from diverse backgrounds in the research enterprise. The NIDCD strives to attract and encourage individuals to consider research careers in the communication sciences at the NIDCD or at NIDCD-supported institutions to enable the research community to be in a position to advance the NIDCD mission and to meet the future health needs of individuals with communication disorders.
Health Disparities Research at the NIDCD
Human communication disorders cross all social and ethnic groups. The NIDCD conducts research to understand the basis of health disparities within its mission areas by determining how communication disorders may contribute to, or be worsened by, differences in health among populations. Recognizing that minorities and individuals with communication disorders are underrepresented in NIDCD-sponsored research and research training activities, the NIDCD is working to increase participation of individuals and groups from diverse backgrounds. Participation of minority or underserved populations in NIDCD-sponsored research advances the NIDCD mission and ensures that everyone benefits from human communication research.
Shared Databases, Registries, and Metrics on Communication Disorders
Biomedical research is rapidly becoming data-intensive as scientists generate and use increasingly large, complex, multidimensional, and diverse datasets. The NIDCD ensures scientific rigor and reproducibility by establishing databases with common measures that encompass the human lifespan for hearing and balance; taste and smell; and voice, speech, and language research. The NIDCD will continue to support data sharing through the development and use of clinical registries, clinical data networks, and other forms of electronic health data to help healthcare providers make evidence-based decisions on best practice and thereby improve outcomes for individuals with communication disorders. The NIDCD is especially committed to developing and implementing infrastructure to identify: 1) investigators with expertise in epidemiology, data registry, clinical trials, and other clinical research and 2) academic- and community-based clinical practice settings with geographical, racial, and ethnic diversity to facilitate rigorous, cost-effective clinical research and maximize human subjects’ protection.
By establishing standard metrics in anatomical, acoustical, and physiological measures, researchers can better define functional communication abilities under real-world conditions. The NIDCD will support new and enhance existing centralized tissue and cell banks to aid access to biological source materials. Standard metrics and centralized tissue banks also help researchers to differentiate clinical subtypes and to identify early preclinical pathology.To improve communication among scientists and clinicians with different specialties, the NIDCD supports development of better measures of performance, communication abilities, disease-specific quality of life instruments, assessment of communication impairments, and outcomes of individuals with communication disorders.
Trans-NIH Efforts Encourage Innovation through Partnerships
While the NIDCD focuses its research efforts on programs that support its mission areas, breakthroughs in related areas, such as neuroscience, genetics, and animal model development, improve our understanding of communication disorders and encourage innovation through partnerships. To support these discoveries, the NIDCD participates in many Trans-NIH initiatives and programs. See Appendix C for examples of Trans-NIH activities.
Excel as a Federal Science Agency by Managing for Results
The NIDCD is a public science agency supported by federal funds. As part of the NIH, the NIDCD is obligated to base its decisions on science, and to make its decision-making process transparent. The NIDCD upholds its accountability to the American public by managing its scientific endeavors with an eye towards achieving results that improve the health of individuals with communication disorders. The NIDCD approaches this responsibility in several different ways, from its reporting as required by a U.S. Law called the Government Performance and Results Act (GPRA), to developing an administrative strategic plan to complement this NIDCD Strategic Plan, and by mitigating the risks involved with administering the NIDCD mission.
GPRA is a U.S. law enacted in 1993. It is designed to improve government performance management, and it requires agencies to manage their performance by setting goals, measuring results, and reporting their progress. To comply with GPRA, the NIH develops an annual plan proposing goals that provide a representative sample of NIH’s activities for each year and describes how these goals will be met, and later in the fiscal year, NIH provides evidence to support any claims for successful achievement of the goals. Each Institute and Center at NIH participates in the GPRA reporting process, including the NIDCD.
The NIDCD’s goal represents only one snapshot of NIDCD’s entire portfolio, but aligns with our Mission to improve the lives of people with communication disorders. The current NIDCD GPRA goal began in FY 2015 and states: By 2020, increase the number of potential treatment options for communication disorders that are being tested in clinical trials by adding one new treatment option per year. To comply with GPRA obligations for this particular goal under the law, the NIDCD proposes a distinct new treatment option that will be tested each fiscal year and then, at the end of that fiscal year, the NIDCD submits evidence that we have tested a new treatment option for a communication disorder. The NIH compiles NIDCD’s annual submission with those from all of the other NIH Institutes and Centers and presents it to the Office of Management and Budget (OMB). OMB includes the NIH information in an annual report on government agency performance that accompanies the President’s annual budget request.
Another way that the NIDCD manages its public funds for results is by developing and using its NIDCD Administrative Strategic Plan. NIDCD staff examine current challenges at the Institute and develop an NIDCD Administrative Strategic Plan to address these challenges. The Plan helps the NIDCD manage its services in support of NIDCD’s mission, and it helps the NIDCD pursue transformative science by:
- Modeling innovative management approaches, encouraging collaboration and the free flow of information, and sharing best practices within and between the NIDCD offices;
- Improving employee quality of life and job satisfaction by implementing clear, consistent, customer-focused service practices;
- Managing services and resources using the principles of efficiency, effectiveness, and quality; and
- Providing better decision-making and transparency by setting goals and then looking back to determine if those goals have been met.
The NIDCD works to ensure that the dollars we invest get results by developing a Risk Management Plan. The plan examines NIDCD’s activities and assesses risks, establishes methods for control of those risks, monitors adherence to the risk-reduction methods, and mitigates risks that are involved with administering the NIDCD mission. The NIDCD plan tries to minimize the risk of failure in all of the NIDCD activities, and it is submitted each year as part of the overall NIH Enterprise Risk Management program.
Future Directions in NIDCD Program Areas
In consultation with communication research scientists and the public, the NIDCD has identified four Priority Areas that have the potential to increase our understanding of the normal and disordered processes of hearing, balance, taste, smell, voice, speech, and language and to further our knowledge in human communication sciences.
Priority Area 1: Understanding Normal Function
Deepen our understanding of the mechanisms underlying normal function of the systems of human communication. By defining what is normal in both animal models and humans, we can better understand mechanisms of disease.
Priority Area 2: Understanding Diseases and Disorders
Increase our knowledge of the mechanisms of diseases, disorders, and dysfunctions that impair human communication and health. Understanding mechanisms that underlie diseases and disorders is an important step in developing better prevention and treatment strategies.
Priority Area 3: Improving Diagnosis, Treatment, and Prevention
Develop, test, and improve diagnosis, treatment, and prevention of diseases, disorders, and dysfunctions of human communication and health. Diagnosis considers normal function and provides targets for prevention and treatment. Improvements in prevention and treatment lead to better outcomes and guide treatment options.
Priority Area 4: Improving Outcomes for Human Communication
Accelerate the translation of research discoveries into practice; increase access to health care; and enhance the delivery, quality, and effectiveness of care to improve personal and public health. Scientifically validated prevention and treatment models will lead to better personal and public health only after adoption into routine practice.
Although the Priority Areas described in this Plan will help the NIDCD identify promising scientific opportunities to advance human communication research over the next five years, the Plan is not meant to be a comprehensive list of all research areas that the NIDCD is currently supporting or plans to support in the future.
The NIDCD will continue to fund as much meritorious research as possible within our program areas of hearing and balance; taste and smell; and voice, speech, and language. Basic and clinical research being supported by the NIDCD will continue to be given high priority. The Institute is committed to supporting new, innovative, hypothesis-driven, meritorious research that can enhance the overall health and quality of life of people with communication disorders.
Hearing and Balance Research
Why the NIDCD Supports Hearing and Balance Research
Loss of hearing or balance negatively impacts quality of life and imposes a significant social and economic burden upon individuals, their families, and the communities in which they live. Millions of Americans experience a hearing or balance disorder at some point in their life, especially as young children or older adults. Common examples include middle-ear infections (otitis media), noise-induced hearing loss, tinnitus, age-related hearing loss, dizziness, and vertigo. Hearing and balance disorders cross all ethnic and socioeconomic lines. Approximately 37.5 million American adults report some degree of hearing loss and 33.4 million adults report a problem during the past 12 months with dizziness or balance, such as vertigo, unsteadiness, or blurred vision after moving the head.1, 2 Among the younger age group, an additional 5.3 percent of American children (3.3 million) also experienced balance and dizziness problems in the last 12 months, as reported by their parents or other adult caregivers.3-6 About two to three of every 1,000 children in the U.S. are born with a detectable level of hearing loss in one or both ears that can affect speech, language, social, and cognitive development. 4, 5 In 2014, one in six U.S. adults aged 18 and older reports trouble hearing without a hearing aid.6
Noise-Induced Hearing Loss
Excess noise is a major contributor to hearing loss in the U.S. Based on nationally representative hearing exam surveys (1999-2004), an estimated 15 percent of Americans aged 20 to 69, or 26 million Americans, reported a history of loud noise exposure and also had high-frequency audiogram results suggesting exposure to excess noise.7 Recent animal studies suggest that noise exposure causing temporary measurable hearing loss may also cause permanent hearing loss that is not readily detectable using standard audiometric testing. Such damage may underlie the common complaint of having difficulty in understanding speech in noisy situations. The NIDCD encourages research to better understand noise-induced auditory damage to inform potential therapies.
Otitis Media
Otitis media (OM), or middle ear infection, is a condition that affects most young children before three years of age. Repeated episodes of OM can contribute to hearing loss and possibly delay language and cognitive skills development. NIDCD-supported research is improving our understanding of susceptibility and pathogenesis of OM. In the future, this research might identify immune pathways to guide effective OM vaccine development.
Age-related Hearing Loss
Age-related hearing loss (presbycusis) is the loss of hearing that gradually occurs during aging. It is one of the most common conditions affecting older and elderly adults with approximately one in three people in the U.S. aged 65 to 74 exhibiting a hearing loss, and nearly half of those older than 75 have difficulty hearing.8 There are many causes of age-related hearing loss. Most commonly, it arises from changes in the inner ear, but it can also result from complex changes along the nerve pathways from the ear to the brain. Understanding the cause of age-related hearing loss and finding ways to prevent it are important research areas supported by the NIDCD.
Tinnitus
Tinnitus, or ringing in the ears, is a disorder that affects approximately 25 million Americans, many of whom also have hearing loss. Severity can range from a mild condition, which requires no intervention, to a severe debilitating disease with significant emotional, social, and economic impact. NIDCD-supported research aims to determine the neural basis of tinnitus, and to develop effective interventions for affected people.
Technology Interventions for Hearing Loss
Individuals with mild-to-severe hearing loss can benefit from using a hearing aid, and many with severe to profound hearing loss benefit from having a cochlear implant. Advances in both hearing aid and cochlear implant technology are improving treatment options for many people with various degrees of hearing loss. For example, individuals may be fitted with hearing aids or cochlear implants on both ears instead of only one ear to improve sound localization and discrimination. In recent years, some people with residual hearing for low-frequency sounds have received both a cochlear implant, to aid them in hearing higher frequency sounds, and a hearing aid to allow them to take advantage of their residual low-frequency hearing. In many cases, this combination (‘hybrid’) strategy results in a significant improvement when listening to speech in background noise.
Animal Models
Animal models of hereditary hearing impairment continue to be instrumental in mapping and cloning many of the gene mutations that contribute to deafness. They help scientists focus on how gene mutations affect protein function and result in deafness, and are a model in which to test therapeutic approaches to treat or prevent hearing loss. These models help us understand the importance of genes in the development and maintenance of the human ear. In addition, mouse and zebrafish models have enabled scientists to examine auditory sensory cells and to characterize the inner ear’s response to sound. Recent research has identified some of the cellular processes that contribute to hair cell damage and death, heralding future studies that may determine the inner ear’s response to mechanical and chemical trauma.
Balance Disorders
The inner ear contains the vestibular system, which includes sensory parts of the inner ear called the vestibular organs. Tiny canals and pouches on both sides of the head are specialized to detect motion and gravity. Their nerve signals interact with other sensory, motor, autonomic and cognitive circuits in the brain for several functions. The vestibular system regulates balanced posture and locomotion, provides spatial and heading orientation for navigation, and stabilizes visual gaze during movement. Normal balance is maintained by integrating inputs from the vestibular, visual, proprioceptive (position sensation), and musculoskeletal systems. Vestibular disorders can lead to dizziness, vertigo, nausea, migraines, blurred vision, and various forms of postural instability. Dysfunctions of the vestibular system can occur independently or with a hearing loss. The NIDCD supports the development of more efficient vestibular testing for improved clinical diagnoses and safer, better tolerated, and more effective treatments for vertigo. NIDCD-supported scientists are also developing vestibular prosthetic devices and minimally invasive surgical techniques to control imbalance and vertigo while preserving hearing and other functions.
The Hearing and Balance Program
The NIDCD Hearing and Balance Program encompasses over half of NIDCD’s research portfolio. To study normal and disordered functions of the auditory and vestibular systems, the NIDCD employs a wide range of research approaches such as molecular genetics, cellular biology, animal models, biomedical imaging, nanotechnology, psychoacoustics, and structural and functional biology. The NIDCD supports research that will lead to improved treatments for, and prevention of, hearing and balance disorders.
Recent Advances in Hearing and Balance Research
Hair Cells
- Scientists have identified TMC1, TMC2, TMHS, and TMIE as proteins important in the conversion of sound-evoked mechanical motion in the inner ear into electric signals to the brain. This knowledge has fundamentally advanced our understanding of how hair cells work.9-15
- High-throughput RNA-sequencing has provided scientists with new insights into the distinct molecular characteristics that occur during the formation of different cell types in the organ of Corti, including hair cells. This information may aid in development of cell-based therapies for treating hearing loss and balance disorders.16-20
- Scientists found that a group of gene regulators called Regulatory Factor Xs (RFXs) helps to drive genes that are preferentially active in hair cells in mice. The researchers concluded that the RFX gene regulators, while not crucial early in the development of hair cells, are necessary for the cells' maturation and long-term survival.21
- Scientists have used proteomics to identify new proteins expressed in hair cell stereociliary bundles. This approach has revealed new insights into hair cell function 22, 23 and identified new components of the hair bundle necessary for hearing and balance.24
Development and Regeneration
- Wnt signaling and Lgr5-expression have been shown to be key for the generation of hair cells in the developing cochlea.25, 26
- Scientists have developed an in vitro technique to turn embryonic stem cells into inner ear hair cells and supporting cells. This technique is well suited for high-throughput screening of drugs for hair cell regeneration.27
- Antisense oligonucleotides have been used to rescue hearing and balance function in a mouse model of human deafness.28
- In the research laboratory, it is now possible to prevent hearing loss and stimulate repair or regenerate sensory cells of the inner ear by transdifferentiating or directly reprogramming cells, or by using gene therapy in animal models.29-31
Hearing Loss
- Damage to spiral ganglion neurons or their synapses in the inner ear may contribute to hearing loss. Scientists have discovered that the synapses between cochlear nerve fibers and inner hair cells are the most vulnerable elements in noise-induced and age-related hearing loss and nerve fibers with high response thresholds are the first to degenerate, which likely contributes to problems with hearing in noisy environments.32-37
- Scientists have determined that unmyelinated type II sensory fibers innervating outer hair cells respond to cellular damage resulting from loud sound and thus may serve as the nociceptors of the inner ear.38, 39
- Dozens of new gene defects responsible for hereditary hearing loss have been identified in recent years, including mutations in the first microRNA (miR-96) involved in hearing loss.40, 41
- The combination of using whole exome sequencing (a technique for sequencing all the expressed genes in a genome) and hearing testing is ushering in a new area of personalized diagnoses, opportunity for earlier intervention, and ultimately, treatment for individuals with hearing loss.42-50
- Gene therapy is being used to correct gene defects that cause hereditary hearing loss and restore auditory function in animal models.51-53
- The use of high-throughput screening in zebrafish is leading to the discovery of new protective compounds that will help diminish or prevent noise- or drug-induced hearing impairment.54-57
- Proof-of-principle studies have shown that small molecules delivered to the cochlea after noise damage can lead to some hair cell regeneration and some functional recovery.58
- Preliminary studies suggest that, in older adults, hearing impairment is associated with cognitive decline, dementia, and depression. Estimated declines are greatest in participants who do not wear a hearing aid. Although data do not currently support a causative relationship, they support future research on causation and potential for reversal with interventions for treatment of hearing loss.59, 60
- Scientists have identified the genetic bases for accelerated age-related hearing loss in humans.61
- Research has shown that genetically producing overexpression of proteins called neurotrophins in the inner ear can elicit regeneration of cochlear synapses after noise damage.62
Otitis Media
- Research has advanced understanding of cell signaling and gene expression patterns of the innate immune system in response to an ear infection (otitis media). 63, 64
- The study of microbial genomes has provided a cost-effective and high-throughput tool to determine genome content of a bacterium that causes ear infections.65
- Scientists have identified and characterized new vaccine candidates with the potential for preventing ear infections.66-68
- To better treat ear infections, scientists have developed a new, noninvasive drug delivery system for the administration of antibiotics and anti-inflammatory agents across the eardrum.69, 70
- Researchers have described how the inflammation induced by bacterial infections treated with aminoglycoside antibiotics potentiates the undesirable side effect of hearing loss.71
Hearing Aids
- Advanced digital technology hearing aids provide noise reduction, directional hearing, and feedback suppression. Binaural hearing aids further improve sound source localization and spatial separation.72
Cochlear Implants and Other Implantable Hearing Devices
- Hybrid devices that combine both electric and acoustic stimulation allow individuals with preserved low-frequency hearing and un-aidable high frequency loss to utilize a combination device that includes a cochlear implant for stimulation of high frequencies and a hearing aid to enhance residual low frequency hearing.73-75
- Scientists are studying further expansion of cochlear implant candidacy in individuals with unilateral deafness who received a cochlear implant. They showed significant improvement in speech perception performance in quiet and in noise after implantation.76 Another study has shown the benefit of cochlear implants in reducing tinnitus in individuals with unilateral hearing loss.77
- More focused electrical stimulation can improve performance for existing cochlear implant users by limiting the overlap between the number of neurons stimulated by different sound frequencies.78, 79
- For individuals in whom cochlear implantation is not an option, auditory brainstem implants now offer an alternative.80
Balance Disorders
- Similar to the benefit of cochlear implants, vestibular implants provide a means of stimulating the afferent nerves within semicircular canals of the inner ear vestibular system. The vestibular prosthesis can mimic the natural vestibular signals 81 to the brain without causing surrounding tissue damage.82 A variety of vestibular disorders can potentially be treated with such a prosthesis.83
Tinnitus
- When cochlear hearing loss occurs, the brain becomes more sensitive to sound to compensate for the reduced peripheral input. Too much sensitivity can make everyday sounds seem too loud (hyperacusis) or can cause ringing in the ear (tinnitus). 84
- Tinnitus and hyperacusis likely involve distributed neural networks that connect multiple brain regions rather than one discrete region. Increased connection and activity between auditory areas of the brain and those associated with emotion, memory, attention, arousal, and spatial location may contribute to some of the maladaptive features of these disorders (e.g., anxiety or fear).85-89
- Improved understanding of the disordered processes that cause tinnitus is leading to better treatments. Animal model studies have identified tinnitus-associated neural changes that commence at the cochlea and extend to more central portions of the brain that process sound. Maladaptive changes in nerve cell behavior likely underlie these changes, resulting in increased spontaneous nerve cell firing rates and synchrony (firing together) among nerve cells in parts of the brain that process sound, possibly resulting in a person “hearing” a sound when no sound stimulus is present. Scientists are currently conducting clinical trials to test the effectiveness of drugs that change the way nerve cells fire to treat acute tinnitus in people. Other new approaches including brain stimulation, such as rTMS (repetitive transcranial magnetic stimulation) 90, hold some promise. Scientists have also had some success with vagal nerve stimulation to eliminate or minimize abnormal nerve cell circuits in individuals with tinnitus. Research has shown that, after cochlear damage, upregulation of somatosensory input to the cochlear nucleus may follow reduction in auditory nerve input, resulting in heightened cochlear nucleus cell responses to somatosensory stimulation. Animals known to have tinnitus have been shown to demonstrate changes in auditory-somatosensory integration, providing a possible mechanism for the treatment of individuals with tinnitus.91, 92
Auditory and Vestibular Processing
- Scientists have been able to determine which speech stimuli cause brain activity by making electrophysiological recordings from electrodes placed on the human brain’s surface. This advance has high significance for the future development of objective ways to measure ability in the parts of the brain that produce and process speech in individuals with normal hearing and hearing impairment.93-95
- Several studies have established that the auditory cortex represents only the sounds of interest and is less affected by the presence of background noise than peripheral auditory neurons in the ears. These findings are crucial for understanding the mechanisms for signal detection in unfavorable listening conditions and the detrimental consequences of even mild hearing loss on those capacities.96-98
- Scientists have made important discoveries to describe the ion channels responsible for transmitting signals to the brain that help us detect our balance and orientation in space.99, 100
- Scientists have integrated their study of auditory and vestibular activity with other sensory systems to advance our understanding of how the nervous system combines and jointly encodes input of sound, sight, and position to improve the ability to orient ourselves with objects around us, while maintaining gaze and posture.101-106
Science Capsule: Balance or Vestibular Disorders in Adults
Balance disorders can result from trauma, disease, or the effects of aging on all the balance-related systems. Vestibular dysfunction can lead to dizziness, vertigo, nausea, migraines, blurred vision, and various forms of postural instability. Episodes of vestibular dizziness or nausea may be relatively brief, but when present can be profoundly disturbing, including disorientation, falling, or even complete incapacitation from physical activity. About 15 percent of American adults (33 million) had a balance or dizziness problem during the past year.2 NIDCD research is supporting the development of more efficient vestibular testing for improved clinical diagnoses and effective pharmacological treatments for vertigo.
A common balance disorder affecting more than one-half million Americans is Ménière’s disease. It can develop at any age, but most often occurs in adults aged 40 to 60. Characteristic symptoms include a combination of vertigo, hearing loss, nausea, tinnitus, and a feeling of fullness in the ear. Ménière’s disease usually affects only one ear. At worst, intense vertigo causes a fall, called a “drop attack,” with possible injury. Because episodes can be repetitive (recurring several times a day, coming and receding over weeks or months) and intense, it can be very debilitating.
Dysfunctions of the vestibular system can occur independently or with a hearing loss, from causes like pharmacotoxicity or head trauma. NIDCD Intramural scientists, at the NIH Clinical Center, evaluate both hearing and vestibular function by testing individuals with and without balance disorders. The goal of the studies is to determine the best way to perform the testing and understand the variations among the test and different individuals. Examples of ongoing research include examining auditory or vestibular function in individuals with neurofibromatosis type 2, Usher syndrome, enlarged vestibular aqueducts, Niemann-Pick type C, xeroderma pigmentosum, and Moebius syndrome.
Balance disorders are associated, as mentioned, with falling, which is the leading cause of injury deaths among older adults. One in three Americans aged 65 and older falls each year 107-110, and falls can result in severe trauma and even loss of life. Each year, more than 4 million older U.S. adults go to emergency departments for fall-related injuries at a cost of $4 billion.111, 112 The NIDCD supports a longitudinal study that measures vestibular function in older adults. The NIDCD is also sponsoring the AVERT (Acute video-oculography for Vertigo in Emergency Rooms for rapid Triage) clinical trial to help diagnose vertigo, dizziness, and other balance problems. The team of researchers is using a diagnostic medical device (video-oculography or VOG) in the triage of patients who go to emergency room with complaints of vertigo and/or dizziness. The device measures abnormal eye movements to differentiate benign causes of the dizziness or imbalance from dangerous causes (like stroke). This study offers the potential for improving standard of care in the diagnosis and treatment of patients with vertigo or dizziness, leading to better outcomes at lower cost.
Priority Areas in Hearing and Balance Research
Priority Area 1: Understanding Normal Function
- Development of the Auditory and Vestibular System: Identify the molecules and the genetic and epigenetic changes involved in development of the peripheral and central auditory and vestibular pathways. Understand how auditory neurons establish tonotopic and other organized sensory representations.
- Homeostasis and Microenvironment: Increase understanding of homeostasis in the inner ear (e.g., ionic composition and maintenance, inflammatory response and toxin elimination, blood-labyrinth barrier, microcirculation, hormonal and other control systems), transport of macromolecules through the round window and in the middle ear (e.g., gas exchange, fluid regulation, innate immunity, and gene expression) and how these homeostatic mechanisms are established developmentally.
- Mechanics: Expand knowledge of three-dimensional mechanics in the cochlea (e.g., interaction of hair cell membranes and stereocilia with supporting structures); in the middle ear (e.g., resolve important issues of middle ear mechanics, including tympanic membrane/ossicular coupling and modes of stapes motion); and in the vestibular system (e.g., cupular and otolithic maintenance of posture and equilibrium).
- Sensory Cell Transduction and Innervation: Identify all the molecular constituents of the hair cell transduction process: nanomechanical properties, molecular motors in hair cell membranes and stereocilia, ion channels and pumps; and their integration for hair cell tuning and maintenance. Identify the factors that promote and maintain hair cell afferent synapses.
- Single Cell Analysis: Define the gene expression profile at the single cell level for multiple different cell types and regions in the cochlea over multiple different time points.
- Functional Connectivity: Clarify how afferent and efferent neural circuits process auditory and vestibular peripheral input. Understand how coding schemes influence plasticity and enable attention, cognition, and stress. Incorporate advanced techniques of functional and structural neural imaging and connectivity, ranging from molecular to systems scale. Bridge non-invasive lower-resolution assessments (imaging and electrophysiological methods – ECoG) of complex sounds (speech) obtained in humans with combined invasive/non-invasive higher-resolution assessments in animal models.
- Perception:
- Auditory System: Determine how sound detection, discrimination, and recognition interact with learning, memory, and attention as well as with vision, tactile sensation, and balance to better understand auditory perception in real-world listening environments, especially in conditions with unfavorable low signal-to-noise ratios.
- Vestibular System: Determine how vestibular, visual, and proprioceptive (the sensing of motion or position) systems interact to perceive space and motion and to maintain orientation.
Priority Area 2: Understanding Diseases and Disorders
- Epidemiology: Investigate natural history; genetic and environmental risk factors; racial, ethnic, and gender differences; and practical objective metrics for subpopulations to inform the development of evidence-based treatment strategies. Explore how complex comorbidities create differences in disease phenotypes and treatment outcomes.
- Genetic Causes of Hearing Loss: Leverage new genetic tools and big data to study genotype and phenotype relationships, e.g., genetic risk factors in noise-induced and age-related hearing loss. Test emerging ideas with animal models using cutting-edge gene-editing technologies (CRISPR). Define the spectrum of genetic contributions to inherited, noise-induced and age-related hearing loss and understand the structural and functional consequences of such mutations. Identify the spectrum of mutations in non-coding sequences that contribute to hereditary hearing loss.
- Single Cell Analysis: Define the gene expression profile at the single cell level for multiple different cell types and regions in the cochlea over multiple different time points in diseased or disordered tissue.
- Otitis Media: Improve understanding of susceptibility and pathogenesis related to genetics, prior upper respiratory infection, eustachian tube dysfunction and reflux, bacterial biofilms and microbiome, polymicrobial infections, dysregulation of innate immunity, inflammation and mucus production, mucosal hyperplasia, and dysregulation of the resolution of inflammation and tissue repair. Define immune pathways for effective middle ear protection by vaccines and for identification of new therapeutic targets. Develop animal models of acute and chronic otitis media. Determine impact of vaccination on disease prevalence and infection by other microbes.
- Inflammatory and Autoimmune Responses of the Inner Ear: Identify and characterize first responders to injury in the inner ear. Determine how molecules and cells cross the blood-labyrinth barriers to initiate immune response and autoimmune disease. Identify genetic and epigenetic risk factors. Investigate innate and cognate immunity in resolution of otitis media.
- Tinnitus and Hyperacusis: Validate assays for tinnitus and hyperacusis in animal models. Couple behavior and neurophysiology in animals to probe mechanisms. Use human brain imaging to identify networks that are involved in tinnitus and hyperacusis.
- Other Acquired Disorders: Improve understanding of the pathogenesis and processes of noise-induced, age-related, traumatic, idiopathic, ototoxic, neurotoxic, metabolic, and hereditary and non-hereditary auditory and vestibular dysfunction. Acquired disorders of interest include Ménière's disease, otosclerosis, idiopathic sudden sensorineural hearing impairment, and the slow hearing decline after hearing-preservation cochlear implantation. Leverage the use of human temporal bones to better understand the clinical progression of disease and disease treatment.
- Pathways and Damage: Determine how the peripheral and central auditory and vestibular pathways are reorganized following injury. Define the long-term changes resulting from sensory cell or neuronal loss. Identify molecular, genetic, and anatomical underpinnings of plasticity in normal and hearing-impaired models. Use human imaging and electrophysiological methods to assess effects of hearing loss on central speech representations. Research the central neural pathways to better understand the relevance of hearing loss to balance disorders.
- Changes in Perception with Disease:
- Auditory System: Identify sources of variance contributing to large individual differences in response to similar intervention strategies among people with hearing loss. Improve understanding of the time course, sensitive periods, and complications of hearing loss across the lifespan. Clarify the aspects of perceptual impairment that are primarily caused by cochlear synaptopathy rather than by cochlear hair cell loss.
- Vestibular System: Understand how disease affects perception of motion and spatial orientation, including connections with limbic and autonomic systems.
Priority Area 3: Improving Diagnosis, Treatment, and Prevention
- Genetic Testing: Improve comprehensive genetic testing by developing more affordable and faster Targeted Genomic Enrichment and Massively Parallel Sequencing Platforms integrating single nucleotide (SNV) and copy number (CNV) variation detection in coding and non-coding regions. Develop better variant annotating and pathogenicity prediction tools.
- Regeneration: Develop in vitro systems to identify genes and factors that promote regeneration of specific cellular phenotypes (e.g., hair cells, supporting cells, spiral ganglion neurons, cells of the stria vascularis); understand factors that promote or inhibit hair cell regeneration spiral ganglion neurite extension and hair cell synaptogenesis; and determine which genes and extracellular factors control cell-specific differentiation.
- Pharmacotherapeutics: Develop targeted delivery of viral vectors for gene therapy and gene repair/correction and site-specific, controlled, sustained molecular therapy for both developing and dysfunctional pathways. Develop therapies to improve neuronal stimulation, resist cell damage, and enhance cell repair. Determine rules governing the diffusion or transport of small molecules, macromolecules, and viruses across the round window membrane.
- Gene Therapy and Gene Delivery: Develop therapies to prevent progression of hearing loss and/or restore function after hearing loss has occurred; identify and catalog viral and non-viral vectors with cell-specific inner ear tropism.
- Tinnitus and Hyperacusis: Apply advanced imaging techniques to provide measures of changed neural activity in people with tinnitus and hyperacusis. Identify pharmacologic agents to prevent tinnitus resulting from traumatic, ototoxic, degenerative, and other acquired disorders. Identify behavioral, pharmacological, surgical, and device-based treatments for improving tinnitus and hyperacusis.
- Otitis Media: Develop new vaccines including polyvalent vaccines for middle ear bacterial and viral infections including polymicrobial infections. Develop new therapeutic agents to enhance innate immunity and host defense, suppress uncontrolled inflammation, mucus production, and tissue repair and speed resolution of inflammation for the treatment of otitis media. Develop new drug delivery systems to the middle ear to treat both middle ear and inner ear diseases.
- Noise-Induced Hearing Loss: Use evidence-based research to develop strategies for preventing noise-induced hearing loss for workers in construction and agriculture and from recreational noise exposure.
- Interventions for Hearing Loss:
- Expand or combine databases for high-resolution molecular, neurophysiological, and psychophysical diagnostics for evidence-based therapeutic approaches.
- Examine existing and develop better aural rehabilitation strategies across the lifespan. Investigate how aural rehabilitation strategies are affected by treating comorbid conditions that influence success, such as co-occurring issues in children with hearing impairment, dementia, or diabetes.
- Improve the performance of traditional (external) hearing aids in background noise and other real-world settings.
- Improve the efficacy of bilateral auditory implants, short electrode implants, and hybrid cochlear implant/hearing aids in the same or opposite ear in conjunction with auditory/aural rehabilitation, assistive devices, and sign language in home and educational environments. Develop alternative means of stimulating the auditory nerve to provide greater channel resolution of auditory implants. Improve prediction of outcome and maintenance of outcome over time.
- Interventions for Dizziness and Balance Disorders: Develop safe and effective pharmacological treatments for vertigo. Develop vestibular prosthetic devices and minimally invasive surgery for better control of imbalance and vertigo while preserving hearing and other functions. Develop improved behavioral approaches for the rehabilitation of chronic vestibular disorders. Develop improved methods of systematic diagnosis and delineation of subtypes of dizziness and vertigo to identify subpopulations that might respond best to targeted therapies. Further research is needed to determine the impact of aural therapies on balance disorders, such as the effect of a cochlear implant or hearing aids on balance function, and the connection with vestibular migraines.
- Management of Infants and Children with Hearing Impairment: Improve early hearing detection and intervention (EHDI) and hearing loss management, including screening, treatment, and rehabilitation. Define the underserved population of infants and children for hearing health care. Determine if early access to hearing health care changes health outcomes later in life. Develop and evaluate the effectiveness of screening methods. Test the effectiveness of various types of intervention strategies.
- Management of Older Adults: Improve hearing loss management, including screening, treatment, and rehabilitation. Define the underserved population of older adults for hearing health care. Determine if early access to hearing health care changes health outcomes later in life. Develop and evaluate the effectiveness of screening methods. Reduce risk of falls in older adults due to imbalance. Develop assistive balance aids, remote sensing feedback devices, and training programs to improve stability and posture in the elderly.
Priority Area 4: Improving Outcomes for Human Communication
- Identifying Impact of Hearing Loss and of Hearing Health Care: Identify factors that influence a person’s motivation and perceived need for hearing health care. Examine the impact of organization, financing, and management of health care services on the delivery, cost, access to, and outcomes of services. Develop innovative delivery systems (e.g., mHealth) to increase awareness, access, and affordability. Identify cost-effective approaches for diagnosis and treatment. Determine the impact of hearing loss on quality of life and general physical and mental health and impact of intervention—including hearing aids and other technologies and communication strategies—on the same outcome measures in real-world environment. In addition, the research recommendations from the 2016 National Academies of Sciences, Engineering, and Medicine report on Hearing Health Care for Adults: Priorities for Improving Access and Affordability continue to be a high priority.
- Auditory Ecology: Use mobile technologies to better understand the real-life listening and communication needs of children and adults with mild to profound hearing loss.
- Comparative Effectiveness Research and Evidence-Based Medicine: Through clinical trials and epidemiological studies, identify best treatments for a given medical condition for a defined set of individuals. Develop and use clinical registries, clinical data networks, and other forms of electronic health data to inform the conscientious, explicit, and judicious use of current best evidence in making decisions about hearing health care options. Develop generalizable quality of life measures that allow us to compete with other health care priorities.
- Implementation and Dissemination Research: Improve implementation of “best practices” among health care providers to translate advances into routine community practice. Increase dissemination of health information to the public to promote healthy behaviors, including the need for intervention in individuals with hearing loss and the dangers of acoustic overexposure to the long-term health of the ear.
- Community-Based Participation in Research: Promote community-based research to identify factors that influence outcomes for people with hearing and balance disorders in diverse real-world settings. Engage deaf and hard of hearing individuals in community-based research to aid in developing behavioral interventions to improve their quality of life. Develop methods to address communication disorders in diverse populations, considering variations in care and practice settings.
Taste and Smell Research
Why the NIDCD Supports Taste and Smell Research
The chemical senses—more commonly known as taste, smell, and chemesthesis (chemically provoked irritation)—enable us to use chemical signals to communicate with the environment and each other. For people, memories of taste and smell experiences are vivid and long lasting, and play an important role in our enjoyment of life. The chemical senses accomplish three major purposes:
- Nutrition: Seeking out safe and nourishing food.
- Protection: Helping us to avoid spoiled food and toxic chemicals.
- Communication: Conveying important information to others.
Specialized cells in the human oral cavity can detect at least five basic taste qualities: sweet, sour, bitter, salty, and savory (umami). Taste cells may also respond to components of fat, to calcium, to complex carbohydrates, and perhaps to other chemical substances found in foods and beverages. Together with the nose, the oral cavity also plays a role in signaling temperature and touch sensations, and in chemesthesis, a multimodal chemical sensitivity of burning sensations that signals the presence of chemical irritants such as capsaicin in hot peppers and toxic chemicals in the air.
Sensory neurons in the nose can detect a wide array of odors, and the sense of smell plays an important role in the perception of food flavor as well. In 1991, Linda Buck and Richard Axel described a very large family of about 1,000 mouse genes that give rise to an equivalent number of olfactory receptor types.113 These receptors are located on olfactory sensory neurons that occupy a small area in the upper part of the nasal epithelium. Drs. Buck and Axel received the 2004 Nobel Prize in Physiology or Medicine for this groundbreaking research, which established a foundation for understanding how odorant molecules interact with their odor receptors.
Each year, more than 200,000 people visit a physician for chemosensory problems such as taste and smell disorders.114 Many more taste and smell disorders go unreported. About 19 percent of U.S. adults aged 40 and older report having had a problem with their ability to taste, and approximately 23 percent report having had a problem with their ability to smell. The likelihood that a person will report a diminished sense of taste and/or smell increases with age. In adults aged 80 and older, nearly 31 percent report a problem with their sense of smell, and more than 27 percent have a problem with their sense of taste.115
Nutrition
The chemical senses are important for regulating food preferences and intake. They evolved to help humans and other animals survive in environments in which required nutrients were scarce and many plants contained poisonous, bitter compounds. Consequently, we seek out sweet, fatty foods and tend to reject the bitterness that characterizes many nutritious vegetables. Although this behavior made sense as humans were evolving, an almost limitless availability of high-calorie foods today can cause the normal function of taste and smell to lead to overconsumption and obesity. More than 2 of every 3 adults are considered to be overweight or obese, and more than 1 of every 3 adults is considered to be obese.116 Individuals who are overweight or obese are at risk of numerous serious conditions (e.g., Type 2 diabetes, heart disease, and sleep apnea).117
People with smell disorders often have problems appreciating the smell of foods and claim that food is less enjoyable. They may change their eating habits, which may have a long-term impact on overall health. Loss of the sense of smell may also cause a person to add too much sugar or salt to make food taste better. This can be a problem for people with certain medical conditions such as diabetes or high blood pressure. In addition, cancer treatments such as radiation and chemotherapy may result in taste and smell loss and an associated decrease in appetite, complicating treatment.
Humans seek out their preferred flavors in foods. Flavor involves interactions between the sensors that signal taste, temperature, touch, smell, and chemesthetic sensations associated with our foods and the parts of the brain that interpret, remember, or think about them. Flavor plays an important role in determining whether someone accepts a particular food and how much of it they choose to eat.118 Scientists studying the chemical senses are interested in learning more about the molecular and developmental bases for how flavors influence food intake and overall health.
Scientists are interested in learning more about how the body detects and responds to salt, fats, and other food characteristics that humans seek out. Data gained from these studies can help us determine new strategies to control overconsumption and improve health without reducing our enjoyment of food. Ongoing research is studying the structure and function of discrete taste, smell, and chemesthetic receptors, as well as their targets within the brain.
Protection
The chemical senses evolved to help us avoid environmental dangers. Bitter tastes warn of potential toxins. Odors associated with spoiled food, toxic volatiles, and dangerous organisms protect us against ingesting or contacting dangerous substances. Odors can even be used to label certain dangerous substances, such as the addition of smelly sulfur compounds to natural gas, which otherwise has no detectable smell. Chemesthesis primarily serves a defensive function, triggering a coughing or gagging reaction that allows us to avoid chemical irritants that cause tissue damage. Loss of chemesthesis results in the inability to detect toxic chemicals in our environment, possibly leading to increased exposure and greater risk of serious health effects. This loss of detection ability persists in people involved in the early rescue, recovery, demolition, or cleanup efforts after the collapse of the World Trade Center towers.119
Communication
Many animals, including mammals, detect chemical communication cues (some of which are called pheromones) given off by animals of the same species. These chemicals convey a variety of messages, including fertility, social rank, health status, and individual identity. Pheromones can also inhibit or induce sexual maturation or mark territory via urination or spraying. Since so many animals use pheromones to communicate information through chemical signals, it seems reasonable to propose that humans do the same. However, the study of chemical communication and pheromones in humans is fraught with controversy. Scientists do not yet agree whether and how humans may use pheromones to communicate. However, other types of odors also affect the way humans interact. For example, people with smell loss may exhibit poor hygiene because they cannot detect their own body odor, thus affecting their normal interactions with others.
Regeneration
The cells that detect chemical signals are constantly renewing and therefore show a remarkable capacity for regeneration. Their locations (in the nose, on the tongue, in the oral cavity) make them susceptible to damage from the environment, so regeneration is required if these cells are to continue to function throughout life. Scientists are interested in learning what enables these tissues to regrow and to re-establish the appropriate connections with the brain. What they learn could be applicable to other human systems and could lead to new treatments for not only taste and smell disorders but also for tissues damaged by stroke or neurodegenerative diseases.
The Taste and Smell Program
The NIDCD Taste and Smell Program supports studies of the chemical senses known as taste, smell, and chemesthesis (chemically provoked irritation) to enhance our understanding of how individuals communicate with their environment and how human chemosensory disorders can be diagnosed and treated. NIDCD-supported research on molecular and cellular biology, animal models, biophysics, and biochemistry of the olfactory and gustatory systems is paving the way for improved diagnosis, prevention, and treatment of chemosensory disorders.
Recent Advances in Taste and Smell Research
Transduction Mechanisms
- The body uses chemosensory transduction mechanisms—processes that enable the conversion of detection into an electrical signal—throughout the oral and nasal cavities. These transduction mechanisms play a major role in the regulation of food intake and the protection of the airways. Scientists have discovered new families of chemosensory receptors (trace amine-associated receptors, formyl peptide receptors) that could detect chemical cues used for communication of odors that signal disease.120
- Scientists have discovered new chemosensory receptors and transduction mechanisms in the gustatory (taste)121-126 and olfactory systems.127, 128
- Scientists are using novel single cell techniques to make numerous copies of the DNA expressed in a single cell as it progresses through early development to explore how olfactory receptor cells choose which receptor to express.129
- Bacteria release quorum signaling molecules to coordinate behaviors such as biofilm formation, virulence, and antibiotic resistance, based on the local density of the bacterial population. Taste receptors expressed in solitary chemosensory cells and ciliated cells of the respiratory epithelium detect irritants and quorum signaling molecules of pathogenic bacteria, evoking protective airway reflexes and inflammatory responses to rid the airways of infection.130, 131
- The use of novel methods132 is rapidly expanding our identification of the ions or molecules (ligands) that bind to a receptor for the diverse set of identified chemosensory receptors.133-136
How Genes and Environment Affect Food Preference
- Experience, internal state, and genetic variation in taste and smell receptor genes affect chemosensory likes and dislikes.137-143 Thus, the chemical senses play key roles in the regulation of food intake that underlies major health issues such as obesity and diabetes.144-146
- The discovery that children and adults experience chemical senses differently has broad implications for the role of flavor in diet selection and health across the lifespan as well as for basic research into the organization and maintenance of chemosensory pathways.147
Chemical Senses and Disease
- Some heritable diseases (e.g., channelopathies and ciliopathies)148-150 as well as neurodegenerative diseases (e.g., Alzheimer’s disease)151-153 have a correlated chemosensory dysfunction that scientists may use to help diagnose diseases or gauge the effectiveness of treatment.
- Individuals who inherit genes that code for one particular version of a bitter taste receptor (a genetic polymorphism) are more susceptible to chronic rhinosinusitis.154 New genetic models of this condition may lead to novel therapeutic interventions for the associated olfactory deficits.155
- Radiation, chemotherapy and traumatic head injuries severely disrupt chemosensory function. Basic research into signaling pathways and transcription factors that regulate development and turnover of chemosensory cells provides a potential basis for restoring chemosensory function.156-161
- Understanding invertebrate chemoreceptor mechanisms and sensitivities162-164 has opened avenues for control and prevention of critical insect-borne diseases such as malaria, dengue fever, encephalitis, and Zika.
Neural Circuitry
- By understanding how taste and smell signaling is set up during normal development, we have a better chance of figuring out how to repair this signaling process if it is damaged. Information about how taste and smell are interpreted in the brain and influence behavior may also be useful for helping us understand why certain tastes and smells make us behave in certain ways, and could help us develop ways to improve mood and modify behavior by modifying this response. Scientists have learned a lot about the cortical circuits that process taste and smell, including:
- Scientists better understand the divisions of function in cortical structures that interpret chemical senses information165-171 and how these circuits fail in pathology.135, 172
- They are learning how cortical circuits create and read odor patterns and the basic circuitry and physiology of these circuits.173-176
- They are using artificial neural networks and optical imaging to define and dissect the circuitry and coding in the chemical senses.177-181
- They have figured out how adult-born neurons can be functionally and synaptically integrated into neural circuits.182
- They have better insight into how activity within these neural circuits translates to chemosensory perception and stimulus identification171, 183-188 and guide such behaviors as emotional response189 and parenting behavior.190
Science Capsule: How Mosquitoes Target their Human Hosts
The NIH and the U.S. Centers for Disease Control and Prevention (CDC) are working to combat the Zika virus, which has achieved pandemic status in South American and the Caribbean. According to the CDC, people become infected with the Zika virus primarily through the bite of infected Aedes aegypti or Aedes albopictus mosquitoes. Zika is spread by the same mosquitoes that spread dengue and chikungunya viruses. The NIDCD supports research projects that focus on mosquitoes because the insects use olfactory cues to target humans and other hosts.
If we determine how certain cues activate mosquito olfactory receptors, we may be able to develop compounds or other methods to block or interfere with this activation and prevent the mosquitoes from detecting humans. An NIDCD-supported scientist found that the domestic form of the A. aegypti mosquito preferentially seeks out human blood over animal blood due to a genetic tweak that makes it more sensitive to human odor.191 Another NIDCD-supported scientist reports that A. aegypti detect plumes of human CO2 upstream and then use visual cues to zero in on human targets.192 Still another group is working to determine the molecular mechanisms by which mosquitoes and other insects seek out moist environments likely to contain human hosts. Scientists now hope to exploit these details to interfere with the insects’ ability to locate human targets.
Another approach to preventing mosquitoes from seeking human hosts is to activate a pathway that prevents mosquitoes from seeking a blood meal. One project in this area is studying the molecules and receptors that are responsible for keeping female mosquitoes from seeking a blood meal for 3 days after a previous meal. If we could simulate these molecule/receptor interactions, we could trick the mosquitoes’ systems into thinking they had already had a meal.
An emergency effort is in progress to assemble the genomic sequence of the A. aegypti mosquito in a matter of months. The goal is to use the genomic information to develop new ways to stop the insects from spreading disease. An NIDCD-supported investigator is leading a group of scientists in this critical project.
Priority Areas in Taste and Smell Research
In developing Priority Areas, the NIDCD took into consideration areas of research that are within the mission of other NIH Institutes, Centers, and Offices (ICO) and are not primarily supported by the NIDCD but that have relevance to the study of chemical senses. These research areas include dietary intake, infectious diseases, and neurological diseases.
- Dietary Intake: The NIDCD supports basic research on chemosensory factors controlling flavor perception, food selection, and related neural pathways. However, research studies that focus exclusively on the consequences of overconsumption or poor diet, including type 2 diabetes, metabolic disorders, stroke, cancer, cardiovascular disease, hypertension, and obesity, are supported by several other NIH ICOs.
- Infectious Diseases: The NIDCD supports studies of basic neural mechanisms of insect olfaction, including olfaction of insects that serve as disease vectors for encephalitis, dengue fever, and malaria. However, the funding of studies focusing exclusively on the infectious nature of these diseases fall outside of NIDCD’s mission area.
- Neurological Diseases: The NIDCD supports studies on alteration and loss of olfactory function, including potential diagnostic significance of such changes, in neurological diseases such as Parkinson's and Alzheimer's disease. However, studies focusing exclusively on causes and treatments of these diseases fall within the mission of areas of other NIH ICOs.
Priority Area 1: Understanding Normal Function
- Fundamental Biology of Chemosensory Function: Continue to develop and apply new tools and approaches to delineate the organization of molecules, cells, and neural circuits underlying the function of the chemesthetic (trigeminal), gustatory and olfactory systems, including development, cell turnover, regeneration, and plasticity.
- Peripheral and Central Bases of Flavor: Understand the complex interactions between peripheral and central aspects of flavor perception, including retronasal or orthonasal olfaction, oral chemesthesis (chemical irritation), taste, oral somesthesis (temperature, texture), memory, and motivational state (such as hunger).
- Sentinel/Sensory Functions: Describe how chemical senses help us avoid dangers such as spoiled or contaminated foods, how they detect potentially toxic chemicals in the environment and in our bodies, and how these protective functions can be damaged and regenerated.
- Genetic Aspects of Chemosensory Sensitivity:
- Genomics: Identify genes involved in the development and normal function of the taste and smell systems, including the use of single-cell profiling approaches.
- Variation: Describe the normal variation in taste and smell sensitivity. Identify the genes involved in order to understand what is outside the range of normal function. Describe how such variation may relate to susceptibility for human communication disorders.
- Experience: Identify genes involved with storing memories of taste and smell. Determine how experience influences future diet.
- Epigenetics: Describe how external factors (e.g., sensory experience, diet, stress) activate and deactivate genes.
- Central Control of Taste and Smell: Characterize top-down control within the central nervous system that modulates sensory input, sensory processing and perception, and determine how such activity may change depending on internal state, motivational or cognitive factors.
- Developing Tools to Measure Taste and Smell Function: Refine, develop, and apply appropriate psychophysical and behavioral methods for assessing taste and smell functions in animal models and humans. Provide practicing physicians with standardized tools to test taste and smell during physical exams or routine office visits. Develop criteria and metrics for the range of “normal” taste and smell by analogy to hearing and vision.
- Develop Novel Approaches to Alter Taste Function: Alter the levels of salt, sugar, and fat intake using innovative methods such as using artificial substitutes or changing learned flavor preferences.
Priority Area 2: Understanding Diseases and Disorders
- Genetic Disorders: Clarify and classify taste and smell disorders caused mainly by significant genetic alterations (e.g., ciliopathies and channelopathies). Determine the normal range of variation of function in the chemical senses as related to genetic polymorphisms.
- Environmental Insults on Taste and Smell: Identify the mechanisms that contribute to taste and smell loss and/or dysfunction resulting from radiation, chemotherapy, head trauma, and toxins.
- Sinusitis/Rhinitis: Identify the molecular and cellular bases for loss of olfaction following nasal cavity or sinus infection, the most common cause of temporary and permanent olfactory loss.
- Understanding How the Activity of the Chemical Senses Can Lead to Excessive Consumption or Malnutrition: Determine whether calorie intake is affected by normal variation or altered function of taste and smell activity.
- Epidemiology: Describe the incidence and prevalence of taste and smell loss and dysfunction. For example, as the population ages, determine how many more people report taste and smell problems that affect quality of life. Enable practical approaches for wider integration of standardized chemosensory measurements into large-scale epidemiological and clinical studies.
Priority Area 3: Improving Diagnosis, Treatment, and Prevention
- Improved Diagnostic Tools and Pharmacological Treatments: Develop and validate tests to evaluate taste and smell function that are practical and affordable for use in the office setting. Develop drugs to treat taste and smell dysfunction, especially drugs which slow apoptosis (cell death) and promote regeneration.
- Regenerative Medicine/Tissue Engineering: Increase understanding of the properties that enable stem cells in the peripheral taste and smell pathways to proliferate and differentiate, providing insights not only for the treatment of taste and smell loss but also for the treatment of other neurological diseases.
- Enhancing the Clinical Enterprise: Promote clinical training in the chemical senses to encourage development of animal models of relevant disorders and promote clinical and translational research, involving interdisciplinary teams of clinicians and basic scientists.
Priority Area 4: Improving Outcomes for Human Communication
- Translational Research: Translational research in the chemical senses is in its infancy, due in part to the modest amount of clinical research that has been conducted. Currently, no evidence-based preventive measures, interventions, or treatments are applied to taste and smell dysfunction. Comparative effectiveness research is premature because of the lack of intervention and treatment strategies and decisions. Because taste and smell loss become increasingly common in a population with a growing proportion of older adults, the NIDCD has identified translational research in the chemical senses as a critical gap area.
Voice, Speech, and Language Research
Why the NIDCD Supports Voice, Speech, and Language Research
Communication allows us to participate in society and is a defining characteristic of what it is to be human. Other organisms clearly communicate; however, in no other species does it appear that communication—specifically the use of language in communication—is as highly developed as in humans, nor as central to an organism’s function and identity. Communication impairments that involve voice, speech, or language often limit a person’s ability to participate in society, whether the activity is educational, occupational, or social. In addition, because effective communication is needed to get aid in life-threatening situations, loss of communication can put people at risk for compromised physical safety and survival.
Human communication requires the brain to integrate complex sensory signals collected by the peripheral organs and to produce neural signals to co-ordinate the muscles involved in speaking and signing language. Human communication systems also rely on the sensory functions of the peripheral organs responsible for hearing, balance, taste, and smell, located in the middle and inner ear, nose, mouth, and throat. They also involve vision (used for sign language and visible speech) and the development of abstract linguistic representations and memory mechanisms, located centrally in the brain. Additionally, communication systems rely on the motor functions of the hands and arms (for sign language and co-speech gesture) and on the peripheral organs of speech production, which include the diaphragm, airway, vocal folds, tongue, lips, and other oral structures.
The interplay between central and peripheral signals, genetics, and environment makes language acquisition a vulnerable process. We don’t understand the causes of many voice, speech, and language disorders, and the path to treatment is often uncertain. Our ability to develop effective treatment is hindered by gaps in evidence for age-appropriate clinical goals, targets of intervention, and expected change trajectories. Researchers are only beginning to understand the developmental course of voice, speech and language markers during childhood that serve as a guide for clinical interventions suited to particular levels of development. In addition, we also need more research on communication problems associated with diseases and disorders most commonly occurring in adults.
While spoken language is the primary way people communicate, it is not the only way. The symbolic nature of language allows us to attribute meaning through not only the voice, speech, language and hearing, but also using visual-manual modes of communication, most notably the use of sign languages and augmentative communication systems. The NIDCD supports research to understand these communication systems, their acquisition and development, and their use when spoken language systems are damaged by trauma or degenerative diseases, or when speech is difficult to acquire due to early hearing loss or injury to the nervous system. This research is also applicable to other human functions because enhanced understanding of visual-manual language systems opens a window into general human cognition.
Developmental Communication Disorders
Nearly 8 percent of children aged 3 to 17 years have had a communication disorder during the past 12 months, according to data from the National Health Interview Survey, 2012.193 In children, delayed speech and language acquisition or impairment are very often significant predictors of future academic, social, vocational, and adaptive outcomes.194-196 These impairments also tend to run in families197, with converging evidence of genetic effects.198 Many communication disorders, such as specific language impairment (SLI) and stuttering, first become apparent when a child normally begins to acquire speech and language. Other developmental disorders may also include communication problems, such as autism spectrum disorder (ASD), Fragile X, or cerebral palsy. One of the hallmarks of ASD is the diminished ability to communicate effectively—particularly in the expression and reception of language. The NIDCD is committed to supporting research efforts to improve the identification speech and language disorders in children and to improve treatments for those disorders.
Language and Literacy
Hearing loss in infancy and childhood may give rise to difficulties in acquiring spoken and written language skills. Children who are deaf are at greater risk for delays in learning to read. Children with normal hearing who have specific language impairment often have reading difficulties upon entry into school. Low proficiency in reading and writing limits job opportunities and economic success. Reading, writing, and communication skills are improving as we add more research on effective ways to teach and address literacy issues in these populations.
Voice and Voice Disorders
About 7.5 million people in the U.S. have trouble using their voice. Vocal fold tissue, a complex biological structure needed for normal voice production, is susceptible to damage from daily insults from environmental pollutants or acid reflux. Such damage may compromise vocal fold integrity over time.199, 200 Laryngeal disorders can cause a significant societal burden due to work-related disability, lost productivity, and direct health care cost (estimated at $11 billion annually).201, 202 The NIDCD supports basic, clinical, and translational research on laryngeal muscle structure and function with respect to normal and disordered voice use, including new prevention and treatment strategies.
Teachers are occupational voice users who represent one of the country’s largest group of employees. Teachers are particularly vulnerable to voice disorders. Between about 11 and 38 percent of teachers have a voice problem on any given day203-205, and cumulative estimates indicate nearly 60 percent of teachers have been affected over their working lives.203 Considering the impact of voice disorders for teachers—the diagnosis, treatment, and substitute teacher costs—the burden to the American economy is substantial, estimated to approach $3 billion annually in 1998.206
Communication Disorders and Neurodegenerative Disorders
Stroke is a leading cause of adult disability in the United States.207 A significant proportion of stroke survivors have communication disorders, such as post-stroke difficulty in using language (aphasia) or difficulty in articulating words (dysarthria) from brain injury. Additionally, neurodegenerative disorders, such as Parkinson’s disease or amyotrophic lateral sclerosis, and injury can lead to impairments in planning and executing motor speech production such as in apraxia or dysarthria. These types of communication problems are a strong predictor of increased isolation and poor quality of life.208 The NIDCD supports research to understand the neurological bases of voice, speech, and language impairments; the correlation of brain imaging data with prognosis; and the development of novel intervention strategies to improve outcomes.
The Voice, Speech, and Language Program
The NIDCD Voice, Speech, and Language program utilizes a wide range of research approaches to develop effective diagnostic and intervention strategies for people with communication impairments. Research in the Voice and Speech area includes studies to determine the nature, causes, treatment, and prevention of disorders of motor speech production throughout the lifespan. The Language area includes the exploration of the genetic bases of child speech and language disorders, as well as characterizing the linguistic and cognitive deficits in children and adults with language disorders.
Recent Advances in Voice, Speech, and Language Research
Transformative Genetic Studies
- Scientists continue to discover new genetic and genomic alterations (including the role of copy number variants) associated with speech and language disorders using new methods such as next-generation whole-exome sequencing.209-213 For example, a new gene, GRIN2A, was identified for focal epilepsies with speech and language disorders, reinforcing an important role for this gene in motor speech function.214, 215 These discoveries are likely to improve the classification, diagnosis, and treatment of speech and language disorders.
- Researchers are learning how reflux from the stomach to the throat and vocal fold tissue harms the larynx. They have demonstrated that reflux significantly alters the expression of 27 genes that are associated with malignant changes of the larynx.216, 217 Understanding how changes in gene expression lead to laryngeal injury provides a comprehensive model for identifying novel diagnostic and therapeutic targets to treat reflux-related injury.
- Researchers generated a transcriptome dataset to capture the complexity of genes responsible for wound healing of the vocal folds. This dataset serves as a resource in developing new studies that would accelerate the identification of novel therapeutic targets to treat reflux-related injury.218
Behavioral Phenotyping
- Studies demonstrated that children with developmental speech and language problems are at a considerable risk for learning disabilities and other psychosocial problems that emerge during adolescence or adulthood.219-221
- Some families with high incidence of stuttering may also have high incidence of other fluency disorders and other speech production difficulties. This finding can lead to new genetic studies across multiple families to define the characteristics of stuttering.222
- Scientists are using new imaging technology to study structural and mechanical characteristics of laryngeal scarring.223 This could provide the foundation for developing improved treatments for one of the most common causes of voice disorders.
- Researchers have identified distinct and viable characteristics of language disorders, extending the research to new populations, such as children who are deaf or minimally verbal children with autism, and to language disorders shared across different populations that may be used in future genetic and treatment studies.224-226 The development of these classification systems will guide future investigations into the genetic, neurologic, and other causal factors that contribute to voice, speech, and language impairments.
Interventions
- Researchers suggest that self-administered computer therapy with single word production improved chronic apraxia of speech. This method shows promise for delivering high-intensity speech and language rehabilitation for individuals recovering from stroke.227
- Scientists have developed a wearable monitoring device to accurately measure voice disorders during daily activities and provide real-time feedback.228, 229 When combined with knowledge of gene expression changes related to vocal fold vibration exposure230, ambulatory monitoring has shown the potential to revolutionize treatment that could facilitate healthier vocal function and enhance diagnosis and treatment options.
- Studies have demonstrated the clinical benefit of speech and language therapy for school-age children who have pragmatic and social communication problems231 and for minimally verbal children with autism.232
- Scientists have extended behavioral treatment research to explore the use of a virtual speech clinician for individuals with aphasia.233 Other studies have shown that spelling therapy combined with supplemental treatments such as transcranial magnetic and direct electrical stimulation of the brain enhances treatment outcomes in individuals with aphasia.234, 235
- Pairing vagus nerve stimulation with a speech sound can improve how the brain processes spoken language.236 These discoveries leverage existing knowledge, inform the development of new treatment paradigms, and improve outcomes for individuals with speech and language disorders.
Bioengineering Advances
- Researchers have expanded the range of augmentative and alternative communication through widely available technologies, such as tablets, for individuals with ASD and related communication disorders.237, 238
- Researchers have developed a model, which can detect and correct speech production errors prior to articulation. This model showed a potential for the development of a brain computer interface (BCI) that uses auditory feedback to allow profoundly paralyzed users to learn to produce speech using a speech synthesizer.239
- Scientists have made significant advances in replacing, engineering, and regenerating vocal fold tissue through the use of stem cells.240 In one study, researchers bioengineered vocal fold tissue using human cells that could produce sound when transplanted into animals.241 Also, investigators have built computational simulations of vocal fold vibrations242-246 that could provide essential information for designing biomaterials that will help restore injured vocal folds. These studies help advance the understanding of normal and disordered vocal function in order to restore vocal fold structure and function and develop improved treatment options.
Imaging Correlations
- Brain imaging technology has identified differences in the white matter of the brain in disorders, such as autism spectrum disorder (ASD) and specific language impairment247, 248, and have demonstrated that common neuropathology tied to shared specific characteristics (e.g., non-word repetition) may be found across different developmental language disorders.249
- Advanced imaging technology has improved our understanding of the complex actions that take place in the part of the brain controlling human speech95 and has allowed for mapping of the functional connections of the brain (connectome) that are responsible for speech control250 and mapping the neural interactions involved in critical elements of the speech motor system.251-254 Similarly, other imaging studies have shown that the brain is organized in specific patterns to perceive speech93, 255-262, including processing vocal tone occurring in the left and right sides of the brain263, and to simultaneously perceive spoken and signed language.264
- Significant advances were made in understanding the anatomical differences of the brain in neurological disorders that impair speech production, such as stuttering265-268 and spasmodic dysphonia.222, 269-271 In addition, scientists can better explain the neural organization of language in a range of acquired language disorders272, 273 and how language networks change as a result of treatment in individuals who have had a stroke.274
- Imaging of the larynx and vocal folds have been refined by ultrasound275 to characterize the relative concentration of collagen and elastic fibers, which are key factors influencing the biomechanical properties of the vocal folds, and by nonlinear laser scanning microscopy and atomic force microscopy-based indentation223 to characterize scarred vocal folds. These imaging techniques are likely to enhance diagnostic capabilities and help evaluate bioengineering techniques used to simulate vocal fold tissue.
Developmental Timing
- Longitudinal studies have documented the predictors and risk factors that are associated with behavior and brain development underlying speech and language in children with or without speech and language disorders.276, 277 This research is now being used to identify early behavioral and neural risk factors that predict later language disorders.278-280
- Studies identified that the quality of caregiver-child interaction is one of the factors that influences how quickly infants process speech.281 Variations in early language experience (early vs. late bilingualism) shape patterns of functional connectivity in the human brain.282 Further, researchers found that the auditory brainstems of adolescents are immature and speech development can be altered.283 Another study helped scientists understand the differences in how the brain perceives vowels and consonants, which may explain some aspects of developmental and acquired speech processing disorders.284
- The first systematic determination of the cellular and molecular progression of vocal fold epithelium development documented five developmental events of the progression from vocal fold initiation in the embryonic anterior foregut tube to fully differentiated and functional adult tissue. The study serves as the necessary foundation for future functional investigations of vocal fold formation.285
- For the first time, a series of high-speed digital imaging studies have compared vocal fold vibration between children and adults. Researchers have demonstrated vocal fold vibration in children is complex and not easily predicted from an adult.286, 287 Further, precise characterization of age-related changes in the larynx paves the way for scientists to design biomaterials with the potential to restore voice to elderly individuals with vocal fold atrophy.288-292
Science Capsule: Spasmodic Dysphonia
Voice production and its quality influence the communicative exchange throughout the lifespan. Voice disorders are overwhelmingly underrecognized. Occupational voice disorders are estimated to affect 28 million Americans and have a significant impact on the livelihood of teachers/professors, TV and radio journalists, lawyers, and singers. The NIDCD supports basic and clinical research studies that focus on normal voice production and the prevention and treatment of voice disorders.
Spasmodic dysphonia (SD), also referred to as laryngeal dystonia, is a voice disorder that belongs to a family of neurological disorders called focal dystonias. SD can affect anyone. When a person with SD attempts to speak, the muscles in the larynx spasm involuntarily and cause the voice to break up and sound strained or breathy. It is a rare disorder, occurring in roughly one to six of every 100,000 people. The first signs of this disorder start to appear in individuals aged 30 to 50 years. More women than men are affected. Currently, there is no cure for SD, and the most common treatment is the injection of very small amounts of botulinum toxin directly into the affected muscles of the larynx. Repeat injections are necessary as the effects last only a few months. In addition, surgical procedures, like the selective laryngeal adductor denervation-reinnervation have yielded good results in people with adductor spasmodic dysphonia. Voice therapy can also be helpful, especially when a patient has developed compensation techniques.
The NIDCD currently funds research to determine the causes and pathophysiology of SD to develop new diagnostics and better treatment options. NIDCD-supported scientists are using multi-modal imaging and next-generation DNA sequencing to identify brain abnormalities and genetic risk factors for SD. By identifying genes responsible for this voice disorder, the Institute is directly addressing the need for better, more accurate detection and diagnosis in this clinical population. NIDCD-supported scientists are now pursuing two new areas for therapies and surgical interventions: locating specific brain areas involved in regulating laryngeal muscles and understanding the neural mechanisms by which they exert their control. In addition, research is also focused on determining if there are deficits in auditory and sensory feedback processing.
The NIDCD will continue to support voice disorders research, guided by recommendations from a 2013 NIDCD-sponsored workshop on voice sciences and disorders. Leading experts in the field agreed that it is essential to strengthen the pipeline of future voice scientists by creating collaborative teams to address lingering research questions. Accordingly, the NIDCD issued two Funding Opportunity Announcements (FOAs) on Advancing Research in Voice Disorders. The initiatives seek cutting-edge research proposals such as the development of biomaterials for engineering vocal fold tissue and development of ambulatory biofeedback approaches for management of patients with voice disorders. Additionally, the FOAs encourage patient outcomes research, health services research, and community-based research with special attention to the needs of individuals with low socio-economic status, disparities, rural, second language populations, and women’s health.
Priority Areas in Voice, Speech, and Language Research
The NIDCD Voice, Speech, and Language Program contains areas of research that overlap with mission areas of other NIH ICOs. In particular:
- Language: NIDCD research focuses on language acquisition in the presence of dysfunctions, diseases, and disorders that alter the traditional developmental course such as hearing loss, ASD, SLI, and aphasia. Research on the normal acquisition of language and on normal language decline from normal aging is within the mission of other NIH ICOs.
- Literacy: As with language, the normal acquisition of literacy skills and individual outcomes in educational settings are within the mission of other NIH ICOs. The NIDCD supports research into literacy for people who are deaf and hard of hearing, the acquisition of written language for people with pre-existing language disorders, improving reading and writing deficits often associated with stroke, and educational interventions to support improved individual outcomes.
- Swallowing: Speech and swallowing functions have shared anatomy, leading the NIDCD to fund some research on swallowing and disordered swallowing (dysphagia). Dysphagia often occurs after head and neck cancer or certain neurological conditions. Many NIH ICOs may have an interest in research on swallowing and dysphagia.
Priority Area 1: Understanding Normal Function
- Modeling: Improve and validate physical, computational, and theoretical modeling of human communication, including vibratory properties of the larynx, neural and speech motor control, and speech language processing.
- Laryngeal System: Examine impact on vocal health from changes to laryngeal muscle function and structure, such as muscle fiber and mucosal changes at the cellular and molecular level. Determine effects from development, the environment, aging, and voice use (voice training and vocal dose—amount, intensity, and distribution).
- Motor Speech Production: Determine the similarities and differences in development and functioning of neural and musculoskeletal systems for human voice and speech production vs. non-speech oral motor control to identify the sensorimotor principles underlying typical speech development and adult speech motor control, and to understand overlapping sensorimotor mechanisms of the larynx.
- Developmental and Neural Plasticity: Identify the developmental course of sensory and motor plasticity and the underlying neural mechanisms associated with voice and speech motor learning in children and adults (e.g., sensorimotor adaptation).
- Sign Language Research: Investigate the acquisition, processing, and neural underpinnings of languages within the visual-manual modality.
- Literacy and Deafness: Identify central and peripheral factors associated with the successful comprehension and use of written language for people who use sign language as their primary way of communication.
Priority Area 2: Understanding Diseases and Disorders
- Natural History and Epidemiology: Identify genetic, neural, sensorimotor, cognitive, linguistic, behavioral, demographic, and environmental factors associated with the progression, developmental course, and long-term outcomes of voice, speech, and language impairments. Determine the relative contribution of those factors to the development of and the recovery from impairment.
- Pathophysiology: Identify the pathophysiologic and cognitive mechanisms underlying both common and rare voice, speech, and language impairments.
- Genetics: Identify genetic and epigenetic factors that contribute to voice, speech, and language impairments, including studies that identify prenatal factors that can modify genetic and epigenetic expression in offspring.
- Developmental and Neural Plasticity: Examine changes in brain structure and functioning in response to behavioral, pathologic, or environmental insult as a basis for voice, speech, and language impairments with an emphasis on developmental timing.
- Co-Occurring Conditions: Examine factors (e.g., social context, inflammatory response, co-morbid conditions) that interact or coexist with primary voice, speech, and language impairments. Examine diagnostic and treatment strategies for voice, speech, and language impairments that may coexist in individuals with deafness, and in individuals with communication disorders. Examine cross-system deficits and their influence on communication health and responsiveness to treatment.
Priority Area 3: Improving Diagnosis, Treatment, and Prevention
- Detection, Diagnosis and Hypothesis-Driven Interventions: Develop biomarkers (e.g., genetic, imaging, behavioral) of objective diagnosis, prognosis, treatment monitoring for developmental and acquired voice, speech, and language impairments. Develop models of intervention informed by cognitive, linguistic, biological, or neurophysiological processes, accounting for cultural and linguistic variation and including predictors of response to treatment. Develop and refine techniques, technology, and instrumentation for improved diagnosis to aid in treatment and prevention.
- Efficacy: Using outcomes-based clinical studies and randomized clinical trials, determine the efficacy of proposed interventions for the prevention and treatment of voice, speech, and language impairments, which can include accounting for cultural and linguistic variation.
- Prevention: Develop and expand programs that prevent the onset or limit the severity of developmental and acquired voice, speech, and language impairments for people with genetic, occupational, environmental, or other risks.
- Understudied Populations: Identify the cause and pathophysiology for understudied populations, such as school-aged children, minimally verbal children with ASD, health disparity groups, and multicultural groups, or understudied conditions, such as stuttering and apraxia of speech in children and adults. Develop methods of assessing and new effective interventions or approaches tailored for understudied populations or conditions.
- Rare Disorders: Develop biomarkers for improved diagnosis, prediction of risk, and treatment response for patients with rare voice, speech and language disorders (e.g., spasmodic dysphonia, paradoxical vocal fold motion).
- Bioengineering, including Assistive Technologies: Harness recent advances in bioengineering to inform the development and evaluate efficacy of wearable monitoring devices, imaging procedures, tissue engineering, bioreactors, and novel augmentative and alternative communication approaches. Enhance brain-computer interface technologies for communication.
- Literacy Skills: Develop methods that promote the acquisition of literacy skills during childhood and improve the reading and writing abilities of people who are deaf and native American Sign Language users.
Priority Area 4: Improving Outcomes for Human Communication
- Novel Delivery: Translate and evaluate efficacy of conventional interventions into new delivery models (e.g., group, family, telehealth, cell-based therapies, and emerging technology platforms).
- Screening: Develop effective and efficient clinical screening tools for use in health and community settings such as schools, primary care physician offices, and senior centers. Develop novel screening tools to document treatment outcomes, to determine communication status, and to improve clinical outcomes in real-world settings. Determine efficacy of screening for improving clinical outcomes.
- Comparative Effectiveness Research and Evidence-Based Medicine: Through clinical trials and epidemiological comparative effectiveness research, identify best treatments for a given communication disorder for a defined set of individuals.
- Patient-Oriented Research: Conduct research to help define the impact of voice, speech, and language communication problems and the desirable/reasonable expectation for quality of life outcome from the individual’s perspective.
- Community-Based Research:
- Promote community-based research and data collection to identify factors that influence outcomes for people with voice, speech, or language impairments, and to inform the development of public policy recommendations.
- Examine community-level health promotion strategies to prevent the occurrence of voice, speech, and language impairments, reduce risk, and improve adherence with treatment.
- Bridging the Gap between Research and Practice: Determine effective dissemination and implementation strategies that enhance the adoption of voice, speech, and language clinical discoveries into routine community practice.
Summary
The mission of the NIDCD is to conduct and support biomedical and behavioral research and research training in the normal and disordered processes of hearing, balance, taste, smell, voice, speech, and language. The Institute also conducts and supports research and research training related to disease prevention and health promotion; addresses special biomedical and behavioral problems associated with people who have communication impairments or disorders; and supports efforts to create devices that substitute for lost and impaired sensory and communication function.
The goals listed in the NIDCD Strategic Plan are an assessment of research areas that present the greatest scientific opportunities and public health needs over the next five years for the three program areas: hearing and balance; taste and smell; and voice, speech, and language. The goals in the Strategic Plan’s Priority Areas are a guide for the following groups:
- Scientists: To better understand the directions that NIDCD research may take in the future;
- The NIDCD: To assist in developing FOAs and to identify projects for HPP nomination; and
- The Public: To understand the state of communication sciences and to discover the scientific breakthroughs that are possible with sustained investments in biomedical research.
The Plan is not a complete list of all research areas that the NIDCD is currently supporting or plans to support in the future. The NIDCD is committed to supporting new, innovative, hypothesis-driven, meritorious research. The Plan will assist us in identifying research areas that have a great opportunity to help the NIDCD improve the health and quality of life of people with communication disorders.
Appendix A: NIDCD Funding History
NIDCD Congressional Appropriations
Appropriated funds for the NIDCD increased dramatically in the first 15 years after the establishment of the Institute in FY 1989. Funding for the NIDCD has remained relatively constant since FY 2005. A notable decrease of approximately 5.2 percent occurred in FY 2013 with the government-wide sequestration, but the FY 2016 appropriation had an increase of 4.4 percent.
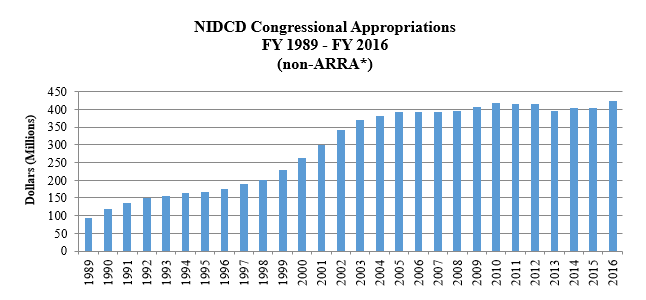 http://officeofbudget.od.nih.gov/approp_hist.html).
http://officeofbudget.od.nih.gov/approp_hist.html).
Total NIDCD Obligated Funds
The NIDCD funds extramural and intramural research in hearing, balance, taste, smell, voice, speech, and language.
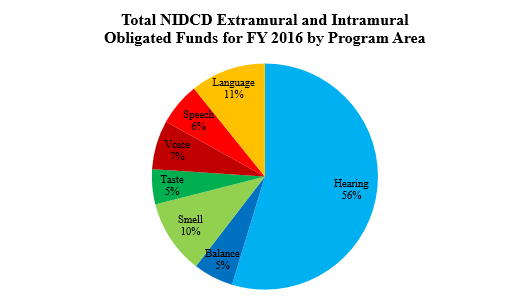
Figure 2: Total NIDCD Extramural and Intramural Obligated Research Funding (excluding ARRA funding and research management and support) for FY 2016. (Data compiled by the NIDCD Financial Management Branch).
Appendix B: The NIDCD 2017-2021 Strategic Plan: The Process
In the fall of 2015, NIDCD’s Science Policy and Planning Branch (SPPB) began the process of updating the current NIDCD Strategic Plan for research, which was expiring in 2016. SPPB took the following steps to update the previous strategic plan:
- Established a Scientific Expert Working Group: In January 2016, the NIDCD identified and invited 12 outside scientific experts to serve on the Working Group to update the NIDCD Strategic Plan. The experts represented NIDCD’s mission areas (Hearing and Balance; Taste and Smell; Voice, Speech, and Language). A roster of the Working Group is below. NIDCD staff also selected a working group chairperson, who is a current NDCD Advisory Council member, and will serve as Council Liaison. SPPB and other NIDCD staff served as resource persons.
- Convened Scientific Expert Working Group via Teleconference: The Working Group held its first conference call in February 2016. SPPB hosted separate working group teleconferences for each of the three main program areas in March 2016. Prior to the teleconferences, working group participants received instructions (including a current NIDCD portfolio analysis, templates with the previous science advances and scientific objectives, and roster/contact information of working group members and NIDCD staff). The Working Group discussed scientific advances that they considered suitable and identified areas of outstanding opportunity and unmet need within their areas of expertise.
- Face to Face Working Group Meeting: The Working Group met in Bethesda, Maryland, at the NIDCD to finalize the draft science advances and research objectives in May 2016.
- Presentation to NDCD Advisory Council: Dr. Charles Liberman, chair of the Working Group, presented the Working Group’s recommendations for research objectives in all three program areas of the NIDCD Plan at the May 20, 2016, meeting. Council members had the opportunity to comment on the draft objectives. In June 2016, the NIDCD SPPB sent the first draft of the Plan to Council for review and comment. At the September 2016 NDCD Advisory Council meeting, NIDCD staff announced that the draft Plan would be made available for public comment.
- Solicited Public Comments: The draft Plan was made available for a 30-day Public Comment Period on the NIDCD website in the fall of 2016. To announce the public comment period, the NIDCD published the Notice (NOT-DC-16-006) in the NIH Guide for Grants and Contracts on September 1, 2016. The NIDCD also published a Notice in the Federal Register on September 1, 2016. The NIDCD received 224 comments from the public.
- Finalized and Posted the Plan on the NIDCD Website: Once appropriate Public Comments were incorporated into the draft approved by NIDCD staff, SPPB finalized the Plan and published it on the NIDCD website in early 2017.
Working Group Roster
Hearing and Balance
| Members | Members |
|---|---|
|
M. Charles Liberman, Ph.D. (Chair) Andrew K. Groves, Ph.D. Jian-Dong Li, M.D., Ph.D. Jay T. Rubinstein, M.D., Ph.D. |
Christoph E. Schreiner, M.D., Ph.D. Richard J. Smith, M.D. Debara L. Tucci, M.D. |
Taste and Smell
| Members | Members |
|---|---|
|
Sue C. Kinnamon, Ph.D. |
Donald A. Wilson, Ph.D. |
Voice, Speech, and Language
| Members | Members |
|---|---|
|
Diane M. Bless, Ph.D. Kristina Simonyan, M.D., Ph.D. |
Helen Tager-Flusberg, Ph.D. |
NIDCD Staff Participants
| Participants | Participants |
|---|---|
|
James F. Battey, Jr., M.D., Ph.D. |
Lisa Kennedy, Ph.D. |
* Note: PDF files require a viewer such as the free Adobe Reader.
Appendix C: NIDCD’s Trans-NIH and Trans-Agency Activities
For the most up-to-date listing of NIDCD’s trans-NIH activities, see https://dpcpsi.nih.gov/collaboration/index.
Trans-NIH Activities
NIH Autism Coordinating Committee (NIH/ACC): Formed by the NIH in 1997 at the request of Congress, the NIH/ACC has been instrumental in planning trans-NIH research initiatives to advance the understanding of autism. The mission is to enhance the quality, pace, and coordination of autism research efforts at the NIH. In addition to program staff from seven ICOs, the NIMH Office of Autism Research Coordination and the NIMH National Database for Autism Research Office participate in NIH/ACC meetings, keeping NIH program offices apprised of their activities and coordinating projects of mutual interest. The NIH/ACC continually monitors the NIH autism research portfolio and the agency's progress toward meeting the goals of the Interagency Autism Coordinating Committee Strategic Plan for ASD Research.
The Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative: Part of a Presidential goal aimed at revolutionizing our understanding of the human brain, the BRAIN Initiative aims to accelerate the development and application of innovative technologies so that researchers will be able to produce a revolutionary new dynamic picture of the brain. For the first time, investigators will be able to show how individual cells and complex neural circuits interact in both time and space. Long desired by researchers seeking new ways to treat, cure, and even prevent brain disorders, this picture will address major gaps in our current knowledge and provide unprecedented opportunities for exploring exactly how the brain enables the human body to record, process, utilize, store, and retrieve vast quantities of information, all at the speed of thought.
National Advisory Board on Medical Rehabilitation Research: The advisory board was stablished by the Director of NIH to advise the directors of NIH ICOs, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), and NICHD's National Center for Medical Rehabilitation Research on matters and policies relating to the Center's programs. The Board is comprised of 12 members representing health and scientific disciplines related to medical rehabilitation and six members representing persons with disabilities.
NIH Medical Rehabilitation Coordinating Committee: Established by the NIH Director to comply with Public Law 101–613, the coordinating committee will make recommendations with respect to the content of the Research Plan and the activities of the NIH Clinical Center that are carried out in conjunction with other components of NIH and with other federal government agencies.
The NIH Human Connectome Project: An ambitious effort to map the neural pathways that underlie human brain function, the Project aims to acquire and share data about the structural and functional connectivity of the human brain. It will greatly advance the capabilities for imaging and analyzing brain connections, resulting in improved sensitivity, resolution, and utility, thereby accelerating progress in the emerging field of human connectomics.
NIH Neuroprosthesis Group: The program officers and staff in this trans-NIH group share an interest in neural prosthetics and neuroengineering research. The group hosts discussions about funding opportunities, meetings, and ongoing projects.
NIH Obesity Research Task Force: The task force was established to accelerate progress in obesity research across NIH in view of the importance of the obesity epidemic as a public health crisis. The task force has been instrumental in fostering trans-NIH collaboration on obesity research, including basic, clinical, and population studies. The task force also sponsors an NIH seminar series on obesity research topics.
Prevention Research Coordinating Committee: The committee serves as a venue for exchanging information on recent scientific advances in disease prevention; examining the impact of new policies on research; planning new or discussing ongoing initiatives; and highlighting program accomplishments. The trans-NIH, trans-agency committee provides a broad perspective on the current state-of-the-science and actively disseminates information about prevention-related activities sponsored by federal and non-federal organizations to the NIH ICOs.
Trans-NIH Rare Diseases Working Group: The working group aims to develop an integrated NIH-wide plan for research in rare diseases that addresses basic, translational, and clinical aspects aimed at the prevention and cure of rare diseases.
Trans-NIH Zebrafish Coordinating Committee: In 1997, the committee was established in response to the scientific community’s recommendation to promote the use of zebrafish as a model organism for the study of vertebrate development and disease. The committee developed a website to provide information about major NIH-organized zebrafish meetings, funding opportunities for zebrafish genomics and genetic resources, major resources generated from grants funded in response to Trans-NIH zebrafish initiatives, training courses and scientific meetings related to the zebrafish initiatives, and selected reports and publications.
Trans-Agency Efforts
Early Hearing Detection and Intervention: The NIDCD’s collaboration with the Centers for Disease Control and Prevention (CDC) and HRSA focuses on a bringing together federal agencies that are interested in issues related to screening infants for hearing loss and providing early intervention.
It’s a Noisy Planet. Protect Their Hearing® Public Education Campaign: The NIDCD sponsors It's a Noisy Planet. Protect Their Hearing®, a national public education campaign to increase awareness among parents of children aged 8 to 12 about the causes and prevention of noise-induced hearing loss. Our federal partner is the National Institute for Occupational Safety and Health (NIOSH) at the CDC, and several nonfederal organizations are involved in the campaign also.
Interagency Autism Coordinating Committee (IACC): Established in accordance with the Autism Collaboration, Accountability, Research, Education, and Support (CARES) Act of 2014, the IACC federal advisory committee is charged with coordinating all efforts with the Department of HHS and across member federal agencies concerning autism spectrum disorder. The committee was established to accelerate progress in autism spectrum disorder biomedical research and services efforts by improving coordination and communication across the federal government and by working in partnership with the autism community.
NIDCD-Supported Epidemiological Studies with the Centers for Disease Control and Prevention (CDC): Several CDC studies are supported by NIDCD.
- NIOSH Audiometric Examinations for Population-Based Surveys: NIDCD provides funding for scientific and technical support as well as quality assurance of three large audiometric examination surveys:
- The National Health and Nutrition Examination Survey (NHANES)
- Age, Gene/Environment Susceptibility Study–Reykjavik Study (AGES–RS)
- The Early Childhood Longitudinal Study
- National Center for Health Statistics (NCHS) Balance/Dizziness Problem Examinations: NIDCD provides funding for the inclusion of Balance/Dizziness Examinations for a representative sample of U.S. adults aged 18 and older and children aged 3 to 17 in the 2016 National Health Interview Survey (NHIS).
- NHIS Hearing Testing: The NIDCD provides funding for a hearing component to the National Health Interview Survey (NHIS) by sponsoring inclusion of many additional questions on hearing loss and tinnitus.
Department of Education Early Childhood Longitudinal Study: The NIDCD supports hearing screening examinations in the Early Childhood Longitudinal Study for the Kindergarten cohort.
Advanced Electrode Microfabrication for Neural Prostheses at the Department of Energy: The NIDCD provides funding support for the Lawrence Livermore National Laboratory to develop precise and rapid construction micromachining techniques and construct arrays of microelectrodes suitable for recording and stimulating neural tissue. These devices will be specifically optimized for use in the NIDCD mission areas of voice, speech, hearing, and balance.
Interagency Committee on Disability Research (ICDR): NIDCD takes part in this government-wide group that meets monthly to discuss issues related to people with disabilities and to coordinate research in this area.
Appendix D: Glossary and Acronym List
Glossary
| Term | Definition |
|---|---|
| afferent |
conducting toward the center; for neurons, conducting nerve impulses toward the spinal cord and brain
|
| aphasia |
total or partial loss of the ability to use or understand language; usually caused by stroke, brain disease, or injury
|
| apraxia of speech |
a speech disorder, also known as verbal apraxia or dyspraxia, in which a person has trouble speaking because of inability to execute a voluntary movement despite normal muscle function
|
| assistive technologies |
products, devices, or equipment that help maintain, increase, or improve the functional capabilities of people with disabilities
|
| auditory nerve |
eighth cranial nerve that connects the inner ear to the brainstem and is responsible for hearing and balance
|
| auditory system |
the outer, middle, and inner ear, along with the neurons and brain regions involved in hearing
|
| autism spectrum disorders |
a spectrum of developmental disorders that begin in early childhood and persists throughout adulthood; autism spectrum disorders affect three crucial areas of development: communication, social interaction, and creative or imaginative play
|
| biofilm |
communities of bacteria, such as the potentially antibiotic-resistant bacterial communities that are present in the middle ears of most children with chronic ear infections
|
| biomarker |
a specific physical trait or a measurable biologically produced change in the body connected with a disease or health condition
|
| chemesthesis |
the “feel” of a chemical; the term describes chemically provoked irritation
|
| chemical senses |
taste and smell
|
| cochlea |
the organ of hearing
|
| cochlear implant |
a medical device that bypasses damaged structures in the inner ear and directly stimulates the auditory nerve, allowing some people who are deaf or hard of hearing to learn to hear and interpret sounds and speech
|
| comorbid |
the existence of one or more co-occurring disorders in addition to a primary disorder
|
| efferent |
conducting away from the center; for neurons, conducting outward from the spinal cord and brain
|
| embryonic stem cells |
cells that are derived from the inner cell mass of blastocyst stage embryos, are capable of dividing without differentiating for a prolonged period in culture, and are known to develop into cells and tissues of the three primary germ layers
|
| epidemiology |
the branch of medical science that investigates all the factors that determine the presence or absence of diseases and disorders in a population
|
| epigenetics |
the study of heritable changes caused by the activation and deactivation of genes without any change in the underlying DNA sequence of the organism
|
| eustachian tube |
a small passageway that connects the upper part of the throat to the middle ear; its job is to supply fresh air to the middle ear, drain fluid, and keep air pressure at a steady level between the nose and the ear
|
| gene expression |
the process by which the information encoded in a gene is used to direct the assembly of a protein molecule; different subsets of genes are expressed in different cell types or under different conditions
|
| genetics |
the study of particular genes, DNA, and heredity
|
| genomics |
the study of the genome (the entire genetic makeup) of an organism
|
| hair cells |
sensory cells of the inner ear, which are topped with hair-like structures (stereocilia) and which transform the mechanical energy of sound waves into nerve impulses
|
| hearing aid |
an electronic device that brings amplified sound to the ear; it usually consists of a microphone, amplifier, and receiver
|
| idiopathic |
relating to a disease or disorder that arises spontaneously or without a known cause
|
| inner ear |
part of the ear that contains both the organ of hearing (the cochlea) and the organ of balance (the labyrinth)
|
| knockout |
an organism that has been genetically engineered to lack one or more specific genes; scientists study knockout organisms to determine the impact of the missing gene, which helps determine its the function
|
| larynx |
valve structure between the trachea (windpipe) and the pharynx (the upper throat) that is the primary organ of voice production
|
| model organism |
animal species used in medical research to mimic aspects of a disease found in humans
|
| mutation |
a change in a DNA sequence that can result from DNA copying mistakes made during cell division, exposure to ionizing radiation, exposure to chemical mutagens, or infection by viruses
|
| neural prostheses |
devices such as the cochlear implant that substitute for an injured or diseased part of the nervous system
|
| nociceptors |
the relatively unspecialized nerve cell endings that initiate the sensation of pain
|
| olfaction |
the sense of smell; to perceive odor or scent through stimuli affecting the olfactory nerves
|
| otitis media |
inflammation of the middle ear caused by infection
|
| ototoxic |
a drug or compound such as a special class of antibiotics, aminoglycoside antibiotics, that can damage the hearing and balance organs located in the inner ear for some individuals
|
| pathogenesis |
the development of a disease or condition, particularly the cellular and molecular origins and causes of disease development
|
| phenotype |
an individual’s physical and behavioral characteristics
|
| pheromone |
chemical substance secreted by an animal that elicits a specific behavioral or physiological response in another animal of the same species
|
| polymorphism |
one of two or more variants of a particular DNA sequence that can correlate with disease, drug response, and other phenotypes; the most common type of polymorphism involves variation at a single base pair (single nucleotide polymorphism) of DNA
|
| psychoacoustics |
the study of sound perception
|
| rhinitis |
inflammation of the mucous membranes of the nose, generally accompanied by discharge (runny nose) and usually caused by a virus infection (e.g., the common cold) or by an allergic reaction (e.g., hay fever)
|
| sinusitis |
inflammation or infection of one of the air-filled nasal sinuses
|
| spasmodic dysphonia |
momentary disruption of voice caused by involuntary movements of one or more muscles of the larynx
|
| spiral ganglion |
the group of nerve cells that serve the sense of hearing by sending a representation of sound from the cochlea to the brain; the cell bodies of the spiral ganglion neurons are found in the spiral structure of the cochlea
|
| stereocilia |
see “hair cells”
|
| stria vascularis |
specialized epithelium lining the cochlear duct that maintains the ion homeostasis of the fluid within the cochlea
|
| stuttering |
a speech disorder in which sounds, syllables, or words are repeated or prolonged, disrupting the normal flow of speech
|
| synapse |
a junction between two nerve cells
|
| tinnitus |
sensation of a ringing, roaring, or buzzing sound in the ears or head when no actual sound stimulus is present in the environment
|
| tonotopic |
the spatial arrangement of where sounds of different frequency are processed in the brain. For example, the auditory nerves that carry signals from adjacent portions of the cochlea project their information to adjacent portions of the auditory cortex
|
| transduction |
the process by which stimuli in the environment are converted into electrical (neural) signals by sensory receptors
|
| vertigo |
illusion of movement; a sensation as if the external world were revolving around an individual (objective vertigo) or as if the individual were revolving in space (subjective vertigo)
|
| vestibular system | system in the body that is responsible for maintaining balance, posture, and the body’s orientation in space; this system also regulates locomotion and other movements and keeps objects in visual focus as the body moves |
Acronymn List
| Acronymn | Definition |
|---|---|
| ACC | Autism Coordinating Committee |
| ARRA | American Recovery and Reinvestment Act |
| ASD | Autism Spectrum Disorder |
| DEA | Division of Extramural Activities |
| DIR | Division of Intramural Research |
| DNA | Deoxyribonucleic Acid |
| DSP | Division of Scientific Programs |
| EHDI | Early Hearing Detection and Intervention |
| FY | Fiscal Year |
| HHS | Department of Health and Human Services |
| HPP | High Program Priority |
| IACC | Interagency Autism Coordinating Committee |
| ICOs | Institutes, Centers, and Offices |
| M.D. | Doctor of Medicine |
| NDCD | National Deafness and Other Communication Disorders |
| NF2 | Neurofibromatosis 2 |
| NICHD | Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| NIDCD | National Institute on Deafness and Other Communication Disorders |
| NIH | National Institutes of Health |
| Ph.D. | Doctor of Philosophy |
| Plan | NIDCD Strategic Plan |
| SLI | Specific Language Impairment |
| SPPB | Science Policy and Planning Branch |
| T2R | Type 2 Taste Receptors |
* Note: PDF files require a viewer such as the free Adobe Reader.
Appendix E: Bibliography
- Blackwell DL, Lucas JW, Clarke TC. Summary health statistics for U.S. adults: National Health Interview Survey, 2012. National Center for Health Statistics. Vital Health Stat 10(260). 2014 Contract No.: 260.
- Ward BK, Agrawal Y, Hoffman HJ, Carey JP, Della Santina CC. Prevalence and impact of bilateral vestibular hypofunction: results from the 2008 US National Health Interview Survey. JAMA Otolaryngol Head Neck Surg. 2013;139(8):803–10. doi: 10.1001/jamaoto.2013.3913. PubMed PMID: 23949355; PMCID: PMC4839981.
- Li CM, Hoffman HJ, Ward BK, Cohen HS, Rine RM. Epidemiology of Dizziness and Balance Problems in Children in the United States: A Population-Based Study. J Pediatr 2016;171:240–7 e3. doi: 10.1016/j.jpeds.2015.12.002. PubMed PMID: 26826885.
- CDC. Identifying infants with hearing loss - United States, 1999–2007. MMWR Morb Mortal Wkly Rep. 2010;59(8):220–3. PubMed PMID: 20203554.
- Vohr B. Overview: Infants and children with hearing loss-part I. Ment Retard Dev Disabil Res Rev. 2003;9(2):62–4. doi: 10.1002/mrdd.10070. PubMed PMID: 12784222.
- Zelaya CE LJ, Hoffman HJ. Self-reported hearing trouble in adults aged 18 and over: United States, 2014. NCHS data brief, no 214. 2015.
- Hoffman HJ, Ko C-W, Themann CL, Dillon CF, Franks JR. Reducing noise-induced hearing loss (NIHL) in adults to achieve U.S. Healthy People 2010 goals. Am J Epidemiol. 2006;(Suppl S); 163(11):S122.
- CDC/NHANES. Based on calculations performed by NIDCD Epidemiology and Statistics Program staff: (1) using data from the 1999–2010 National Health and Nutrition Examination Survey (NHANES); (2) applying the definition of disabling hearing loss used by the 2010 Global Burden of Disease Expert Hearing Loss Team (hearing loss of 35 decibels or more in the better ear, the level at which adults could generally benefit from hearing aids) 2010.
- Kawashima Y, Geleoc GS, Kurima K, Labay V, Lelli A, Asai Y, Makishima T, Wu DK, Della Santina CC, Holt JR, Griffith AJ. Mechanotransduction in mouse inner ear hair cells requires transmembrane channel-like genes. J Clin Invest. 2011;121(12):4796–809. doi: 10.1172/JCI60405. PubMed PMID: 22105175; PMCID: PMC3223072.
- Kurima K, Ebrahim S, Pan B, Sedlacek M, Sengupta P, Millis BA, Cui R, Nakanishi H, Fujikawa T, Kawashima Y, Choi BY, Monahan K, Holt JR, Griffith AJ, Kachar B. TMC1 and TMC2 Localize at the Site of Mechanotransduction in Mammalian Inner Ear Hair Cell Stereocilia. Cell Rep. 2015;12(10):1606–17. doi: 10.1016/j.celrep.2015.07.058. PubMed PMID: 26321635; PMCID: PMC4569002.
- Liedtke W. A precisely defined role for the tip link-associated protein TMIE in the mechanoelectrical transduction channel complex of inner ear hair cells. Neuron. 2014;84(5):889–91. doi: 10.1016/j.neuron.2014.11.015. PubMed PMID: 25475183.
- Maeda R, Kindt KS, Mo W, Morgan CP, Erickson T, Zhao H, Clemens-Grisham R, Barr-Gillespie PG, Nicolson T. Tip-link protein protocadherin 15 interacts with transmembrane channel-like proteins TMC1 and TMC2. Proc Natl Acad Sci U S A. 2014;111(35):12907–12. doi: 10.1073/pnas.1402152111. PubMed PMID: 25114259; PMCID: PMC4156717.
- Pan B, Geleoc GS, Asai Y, Horwitz GC, Kurima K, Ishikawa K, Kawashima Y, Griffith AJ, Holt JR. TMC1 and TMC2 are components of the mechanotransduction channel in hair cells of the mammalian inner ear. Neuron. 2013;79(3):504–15. doi: 10.1016/j.neuron.2013.06.019. PubMed PMID: 23871232; PMCID: PMC3827726.
- Zhao B, Wu Z, Grillet N, Yan L, Xiong W, Harkins-Perry S, Muller U. TMIE is an essential component of the mechanotransduction machinery of cochlear hair cells. Neuron. 2014;84(5):954–67. doi: 10.1016/j.neuron.2014.10.041. PubMed PMID: 25467981; PMCID: PMC4258123.
- Xiong W, Grillet N, Elledge HM, Wagner TF, Zhao B, Johnson KR, Kazmierczak P, Muller U. TMHS is an integral component of the mechanotransduction machinery of cochlear hair cells. Cell. 2012;151(6):1283–95. doi: 10.1016/j.cell.2012.10.041. PubMed PMID: 23217710; PMCID: PMC3522178.
- Burns JC, Kelly MC, Hoa M, Morell RJ, Kelley MW. Single-cell RNA-Seq resolves cellular complexity in sensory organs from the neonatal inner ear. Nat Commun. 2015;6:8557. doi: 10.1038/ncomms9557. PubMed PMID: 26469390; PMCID: PMC4634134.
- Cai T, Jen HI, Kang H, Klisch TJ, Zoghbi HY, Groves AK. Characterization of the transcriptome of nascent hair cells and identification of direct targets of the Atoh1 transcription factor. J Neurosci. 2015;35(14):5870–83. doi: 10.1523/JNEUROSCI.5083–14.2015. PubMed PMID: 25855195; PMCID: PMC4388939.
- Scheffer DI, Shen J, Corey DP, Chen ZY. Gene Expression by Mouse Inner Ear Hair Cells during Development. J Neurosci. 2015;35(16):6366–80. doi: 10.1523/JNEUROSCI.5126–14.2015. PubMed PMID: 25904789; PMCID: PMC4405555.
- Shen J, Scheffer DI, Kwan KY, Corey DP. SHIELD: an integrative gene expression database for inner ear research. Database (Oxford). 2015;2015:bav071. doi: 10.1093/database/bav071. PubMed PMID: 26209310; PMCID: PMC4513695.
- Waldhaus J, Durruthy-Durruthy R, Heller S. Quantitative High-Resolution Cellular Map of the Organ of Corti. Cell Rep. 2015;11(9):1385–99. doi: 10.1016/j.celrep.2015.04.062. PubMed PMID: 26027927; PMCID: PMC4465070.
- Elkon R, Milon B, Morrison L, Shah M, Vijayakumar S, Racherla M, Leitch CC, Silipino L, Hadi S, Weiss-Gayet M, Barras E, Schmid CD, Ait-Lounis A, Barnes A, Song Y, Eisenman DJ, Eliyahu E, Frolenkov GI, Strome SE, Durand B, Zaghloul NA, Jones SM, Reith W, Hertzano R. RFX transcription factors are essential for hearing in mice. Nat Commun. 2015;6:8549. doi: 10.1038/ncomms9549. PubMed PMID: 26469318; PMCID: PMC4634137.
- Shin JB, Krey JF, Hassan A, Metlagel Z, Tauscher AN, Pagana JM, Sherman NE, Jeffery ED, Spinelli KJ, Zhao H, Wilmarth PA, Choi D, David LL, Auer M, Barr-Gillespie PG. Molecular architecture of the chick vestibular hair bundle. Nat Neurosci. 2013;16(3):365–74. doi: 10.1038/nn.3312. PubMed PMID: 23334578; PMCID: PMC3581746.
- Wilmarth PA, Krey JF, Shin JB, Choi D, David LL, Barr-Gillespie PG. Hair-bundle proteomes of avian and mammalian inner-ear utricles. Sci Data. 2015;2:150074. doi: 10.1038/sdata.2015.74. PubMed PMID: 26645194; PMCID: PMC4672683.
- Francis SP, Krey JF, Krystofiak ES, Cui R, Nanda S, Xu W, Kachar B, Barr-Gillespie PG, Shin JB. A short splice form of Xin-actin binding repeat containing 2 (XIRP2) lacking the Xin repeats is required for maintenance of stereocilia morphology and hearing function. J Neurosci. 2015;35(5):1999–2014. doi: 10.1523/JNEUROSCI.3449–14.2015. PubMed PMID: 25653358; PMCID: PMC4315831.
- Chai R, Kuo B, Wang T, Liaw EJ, Xia A, Jan TA, Liu Z, Taketo MM, Oghalai JS, Nusse R, Zuo J, Cheng AG. Wnt signaling induces proliferation of sensory precursors in the postnatal mouse cochlea. Proc Natl Acad Sci U S A. 2012;109(21):8167–72. doi: 10.1073/pnas.1202774109. PubMed PMID: 22562792; PMCID: PMC3361451.
- Shi F, Kempfle JS, Edge AS. Wnt-responsive Lgr5-expressing stem cells are hair cell progenitors in the cochlea. J Neurosci. 2012;32(28):9639–48. doi: 10.1523/JNEUROSCI.1064–12.2012. PubMed PMID: 22787049; PMCID: PMC3417821.
- Koehler KR, Hashino E. 3D mouse embryonic stem cell culture for generating inner ear organoids. Nat Protoc. 2014;9(6):1229–44. doi: 10.1038/nprot.2014.100. PubMed PMID: 24784820.
- Lentz JJ, Jodelka FM, Hinrich AJ, McCaffrey KE, Farris HE, Spalitta MJ, Bazan NG, Duelli DM, Rigo F, Hastings ML. Rescue of hearing and vestibular function by antisense oligonucleotides in a mouse model of human deafness. Nat Med. 2013;19(3):345–50. doi: 10.1038/nm.3106. PubMed PMID: 23380860; PMCID: PMC3657744.
- Bermingham-McDonogh O, Reh TA. Regulated reprogramming in the regeneration of sensory receptor cells. Neuron. 2011;71(3):389–405. doi: 10.1016/j.neuron.2011.07.015. PubMed PMID: 21835338; PMCID: PMC4403668.
- Conde de Felipe MM, Feijoo Redondo A, Garcia-Sancho J, Schimmang T, Duran Alonso MB. Cell- and gene-therapy approaches to inner ear repair. Histol Histopathol. 2011;26(7):923–40. PubMed PMID: 21630222.
- Shibata SB, Ranum PT, Moteki H, Pan B, Goodwin AT, Goodman SS, Abbas PJ, Holt JR, Smith RJ. RNA Interference Prevents Autosomal-Dominant Hearing Loss. Am J Hum Genet. 2016. doi: 10.1016/j.ajhg.2016.03.028. PubMed PMID: 27236922.
- Hickox AE, Liberman MC. Is noise-induced cochlear neuropathy key to the generation of hyperacusis or tinnitus? J Neurophysiol. 2014;111(3):552–64. doi: 10.1152/jn.00184.2013. PubMed PMID: 24198321; PMCID: PMC3921399.
- Kujawa SG, Liberman MC. Synaptopathy in the noise-exposed and aging cochlea: Primary neural degeneration in acquired sensorineural hearing loss. Hear Res. 2015;330(Pt B):191–9. doi: 10.1016/j.heares.2015.02.009. PubMed PMID: 25769437; PMCID: PMC4567542.
- Liberman MC. Hidden Hearing Loss. Sci Am. 2015;313(2):48–53. PubMed PMID: 26349143.
- Sergeyenko Y, Lall K, Liberman MC, Kujawa SG. Age-related cochlear synaptopathy: an early-onset contributor to auditory functional decline. J Neurosci. 2013;33(34):13686–94. doi: 10.1523/JNEUROSCI.1783–13.2013. PubMed PMID: 23966690; PMCID: PMC3755715.
- Furman AC, Kujawa SG, Liberman MC. Noise-induced cochlear neuropathy is selective for fibers with low spontaneous rates. J Neurophysiol. 2013;110(3):577–86. doi: 10.1152/jn.00164.2013. PubMed PMID: 23596328; PMCID: PMC3742994.
- Viana LM, O'Malley JT, Burgess BJ, Jones DD, Oliveira CA, Santos F, Merchant SN, Liberman LD, Liberman MC. Cochlear neuropathy in human presbycusis: Confocal analysis of hidden hearing loss in post-mortem tissue. Hear Res. 2015;327:78–88. doi: 10.1016/j.heares.2015.04.014. PubMed PMID: 26002688; PMCID: PMC4554812.
- Flores EN, Duggan A, Madathany T, Hogan AK, Marquez FG, Kumar G, Seal RP, Edwards RH, Liberman MC, Garcia-Anoveros J. A non-canonical pathway from cochlea to brain signals tissue-damaging noise. Curr Biol. 2015;25(5):606–12. doi: 10.1016/j.cub.2015.01.009. PubMed PMID: 25639244; PMCID: PMC4348215.
- Liu C, Glowatzki E, Fuchs PA. Unmyelinated type II afferent neurons report cochlear damage. Proc Natl Acad Sci U S A. 2015;112(47):14723–7. doi: 10.1073/pnas.1515228112. PubMed PMID: 26553995; PMCID: PMC4664349.
- Kuhn S, Johnson SL, Furness DN, Chen J, Ingham N, Hilton JM, Steffes G, Lewis MA, Zampini V, Hackney CM, Masetto S, Holley MC, Steel KP, Marcotti W. miR-96 regulates the progression of differentiation in mammalian cochlear inner and outer hair cells. Proc Natl Acad Sci U S A. 2011;108(6):2355–60. doi: 10.1073/pnas.1016646108. PubMed PMID: 21245307; PMCID: PMC3038748.
- Mencia A, Modamio-Hoybjor S, Redshaw N, Morin M, Mayo-Merino F, Olavarrieta L, Aguirre LA, del Castillo I, Steel KP, Dalmay T, Moreno F, Moreno-Pelayo MA. Mutations in the seed region of human miR-96 are responsible for nonsyndromic progressive hearing loss. Nat Genet. 2009;41(5):609–13. doi: 10.1038/ng.355. PubMed PMID: 19363479.
- Shearer AE, DeLuca AP, Hildebrand MS, Taylor KR, Gurrola J, 2nd, Scherer S, Scheetz TE, Smith RJ. Comprehensive genetic testing for hereditary hearing loss using massively parallel sequencing. Proc Natl Acad Sci U S A. 2010;107(49):21104–9. doi: 10.1073/pnas.1012989107. PubMed PMID: 21078986; PMCID: PMC3000272.
- Sloan-Heggen CM, Babanejad M, Beheshtian M, Simpson AC, Booth KT, Ardalani F, Frees KL, Mohseni M, Mozafari R, Mehrjoo Z, Jamali L, Vaziri S, Akhtarkhavari T, Bazazzadegan N, Nikzat N, Arzhangi S, Sabbagh F, Otukesh H, Seifati SM, Khodaei H, Taghdiri M, Meyer NC, Daneshi A, Farhadi M, Kahrizi K, Smith RJ, Azaiez H, Najmabadi H. Characterising the spectrum of autosomal recessive hereditary hearing loss in Iran. J Med Genet. 2015;52(12):823–9. doi: 10.1136/jmedgenet–2015–103389. PubMed PMID: 26445815; PMCID: PMC4733363.
- Alford RL, Arnos KS, Fox M, Lin JW, Palmer CG, Pandya A, Rehm HL, Robin NH, Scott DA, Yoshinaga-Itano C, Loss AWGoUoGEGftEDoCH, Professional P, Guidelines C. American College of Medical Genetics and Genomics guideline for the clinical evaluation and etiologic diagnosis of hearing loss. Genet Med. 2014;16(4):347–55. doi: 10.1038/gim.2014.2. PubMed PMID: 24651602.
- Kimberling WJ, Hildebrand MS, Shearer AE, Jensen ML, Halder JA, Trzupek K, Cohn ES, Weleber RG, Stone EM, Smith RJ. Frequency of Usher syndrome in two pediatric populations: Implications for genetic screening of deaf and hard of hearing children. Genet Med. 2010;12(8):512–6. doi: 10.1097/GIM.0b013e3181e5afb8. PubMed PMID: 20613545; PMCID: PMC3131500.
- Declau F, Boudewyns A, Van den Ende J, Peeters A, van den Heyning P. Etiologic and audiologic evaluations after universal neonatal hearing screening: analysis of 170 referred neonates. Pediatrics. 2008;121(6):1119–26. doi: 10.1542/peds.2007–1479. PubMed PMID: 18519481.
- Choi BY, Park G, Gim J, Kim AR, Kim BJ, Kim HS, Park JH, Park T, Oh SH, Han KH, Park WY. Diagnostic application of targeted resequencing for familial nonsyndromic hearing loss. PLoS One. 2013;8(8):e68692. doi: 10.1371/journal.pone.0068692. PubMed PMID: 23990876; PMCID: PMC3750053.
- Vona B, Muller T, Nanda I, Neuner C, Hofrichter MA, Schroder J, Bartsch O, Lassig A, Keilmann A, Schraven S, Kraus F, Shehata-Dieler W, Haaf T. Targeted next-generation sequencing of deafness genes in hearing-impaired individuals uncovers informative mutations. Genet Med. 2014;16(12):945–53. doi: 10.1038/gim.2014.65. PubMed PMID: 24875298; PMCID: PMC4262760.
- Jayawardena AD, Shearer AE, Smith RJ. Sensorineural Hearing Loss: A Changing Paradigm for Its Evaluation. Otolaryngol Head Neck Surg. 2015;153(5):843–50. doi: 10.1177/0194599815596727. PubMed PMID: 26216887; PMCID: PMC4730883.
- Sloan-Heggen CM, Bierer AO, Shearer AE, Kolbe DL, Nishimura CJ, Frees KL, Ephraim SS, Shibata SB, Booth KT, Campbell CA, Ranum PT, Weaver AE, Black-Ziegelbein EA, Wang D, Azaiez H, Smith RJ. Comprehensive genetic testing in the clinical evaluation of 1119 patients with hearing loss. Hum Genet. 2016;135(4):441–50. doi: 10.1007/s00439–016–1648–8. PubMed PMID: 26969326; PMCID: PMC4796320.
- Akil O, Seal RP, Burke K, Wang C, Alemi A, During M, Edwards RH, Lustig LR. Restoration of hearing in the VGLUT3 knockout mouse using virally mediated gene therapy. Neuron. 2012;75(2):283–93. doi: 10.1016/j.neuron.2012.05.019. PubMed PMID: 22841313; PMCID: PMC3408581.
- Askew C, Rochat C, Pan B, Asai Y, Ahmed H, Child E, Schneider BL, Aebischer P, Holt JR. Tmc gene therapy restores auditory function in deaf mice. Sci Transl Med. 2015;7(295):295ra108. doi: 10.1126/scitranslmed.aab1996. PubMed PMID: 26157030.
- Di Domenico M, Ricciardi C, Martone T, Mazzarella N, Cassandro C, Chiarella G, D'Angelo L, Cassandro E. Towards gene therapy for deafness. J Cell Physiol. 2011;226(10):2494–9. doi: 10.1002/jcp.22617. PubMed PMID: 21792906.
- Ou HC, Keating S, Wu P, Simon JA, Raible DW, Rubel EW. Quinoline ring derivatives protect against aminoglycoside-induced hair cell death in the zebrafish lateral line. J Assoc Res Otolaryngol. 2012;13(6):759–70. doi: 10.1007/s10162–012–0353–0. PubMed PMID: 23053627; PMCID: PMC3505584.
- Ou H, Simon JA, Rubel EW, Raible DW. Screening for chemicals that affect hair cell death and survival in the zebrafish lateral line. Hear Res. 2012;288(1–2):58–66. doi: 10.1016/j.heares.2012.01.009. PubMed PMID: 22310494; PMCID: PMC3371178.
- Stawicki TM, Esterberg R, Hailey DW, Raible DW, Rubel EW. Using the zebrafish lateral line to uncover novel mechanisms of action and prevention in drug-induced hair cell death. Front Cell Neurosci. 2015;9:46. doi: 10.3389/fncel.2015.00046. PubMed PMID: 25741241; PMCID: PMC4332341.
- Thomas AJ, Wu P, Raible DW, Rubel EW, Simon JA, Ou HC. Identification of small molecule inhibitors of cisplatin-induced hair cell death: results of a 10,000 compound screen in the zebrafish lateral line. Otol Neurotol. 2015;36(3):519–25. doi: 10.1097/MAO.0000000000000487. PubMed PMID: 25687728; PMCID: PMC4332566.
- Mizutari K, Fujioka M, Hosoya M, Bramhall N, Okano HJ, Okano H, Edge AS. Notch inhibition induces cochlear hair cell regeneration and recovery of hearing after acoustic trauma. Neuron. 2013;77(1):58–69. doi: 10.1016/j.neuron.2012.10.032. PubMed PMID: 23312516; PMCID: PMC3573859.
- Deal JA, Sharrett AR, Albert MS, Coresh J, Mosley TH, Knopman D, Wruck LM, Lin FR. Hearing impairment and cognitive decline: a pilot study conducted within the atherosclerosis risk in communities neurocognitive study. Am J Epidemiol. 2015;181(9):680–90. doi: 10.1093/aje/kwu333. PubMed PMID: 25841870; PMCID: PMC4408947.
- Whitson HE, Lin FR. Hearing and vision care for older adults: sensing a need to update Medicare policy. JAMA. 2014;312(17):1739–40. doi: 10.1001/jama.2014.13535. PubMed PMID: 25369486; PMCID: PMC4260264.
- Van Laer L, Huyghe JR, Hannula S, Van Eyken E, Stephan DA, Maki-Torkko E, Aikio P, Fransen E, Lysholm-Bernacchi A, Sorri M, Huentelman MJ, Van Camp G. A genome-wide association study for age-related hearing impairment in the Saami. Eur J Hum Genet. 2010;18(6):685–93. doi: 10.1038/ejhg.2009.234. PubMed PMID: 20068591; PMCID: PMC2987344.
- Wan G, Gomez-Casati ME, Gigliello AR, Liberman MC, Corfas G. Neurotrophin-3 regulates ribbon synapse density in the cochlea and induces synapse regeneration after acoustic trauma. Elife. 2014;3. doi: 10.7554/eLife.03564. PubMed PMID: 25329343; PMCID: PMC4227045.
- Kurabi A, Pak K, Ryan AF, Wasserman SI. Innate Immunity: Orchestrating Inflammation and Resolution of Otitis Media. Curr Allergy Asthma Rep. 2016;16(1):6. doi: 10.1007/s11882–015–0585–2. PubMed PMID: 26732809.
- Kerschner JE, Hong W, Taylor SR, Kerschner JA, Khampang P, Wrege KC, North PE. A novel model of spontaneous otitis media with effusion (OME) in the Oxgr1 knock-out mouse. Int J Pediatr Otorhinolaryngol. 2013;77(1):79–84. doi: 10.1016/j.ijporl.2012.09.037. PubMed PMID: 23200873; PMCID: PMC3535456.
- Eutsey RA, Hiller NL, Earl JP, Janto BA, Dahlgren ME, Ahmed A, Powell E, Schultz MP, Gilsdorf JR, Zhang L, Smith A, Murphy TF, Sethi S, Shen K, Post JC, Hu FZ, Ehrlich GD. Design and validation of a supragenome array for determination of the genomic content of Haemophilus influenzae isolates. BMC Genomics. 2013;14:484. doi: 10.1186/1471–2164–14–484. PubMed PMID: 23865594; PMCID: PMC3723446.
- Novotny LA, Jurcisek JA, Ward MO, Jr., Jordan ZB, Goodman SD, Bakaletz LO. Antibodies against the majority subunit of type IV Pili disperse nontypeable Haemophilus influenzae biofilms in a LuxS-dependent manner and confer therapeutic resolution of experimental otitis media. Mol Microbiol. 2015;96(2):276–92. doi: 10.1111/mmi.12934. PubMed PMID: 25597921; PMCID: PMC4423401.
- Murphy TF. Vaccines for Nontypeable Haemophilus influenzae: the Future Is Now. Clin Vaccine Immunol. 2015;22(5):459–66. doi: 10.1128/CVI.00089–15. PubMed PMID: 25787137; PMCID: PMC4412935.
- Yildirim I, Shea KM, Pelton SI. Pneumococcal Disease in the Era of Pneumococcal Conjugate Vaccine. Infect Dis Clin North Am. 2015;29(4):679–97. doi: 10.1016/j.idc.2015.07.009. PubMed PMID: 26610421; PMCID: PMC4662776.
- Khoo X, Simons EJ, Chiang HH, Hickey JM, Sabharwal V, Pelton SI, Rosowski JJ, Langer R, Kohane DS. Formulations for trans-tympanic antibiotic delivery. Biomaterials. 2013;34(4):1281–8. doi: 10.1016/j.biomaterials.2012.10.025. PubMed PMID: 23146430; PMCID: PMC3511665.
- Lee JY, Komatsu K, Lee BC, Miyata M, O'Neill Bohn A, Xu H, Yan C, Li JD. Vinpocetine inhibits Streptococcus pneumoniae-induced upregulation of mucin MUC5AC expression via induction of MKP-1 phosphatase in the pathogenesis of otitis media. J Immunol. 2015;194(12):5990–8. doi: 10.4049/jimmunol.1401489. PubMed PMID: 25972475; PMCID: PMC4456630.
- Koo JW, Quintanilla-Dieck L, Jiang M, Liu J, Urdang ZD, Allensworth JJ, Cross CP, Li H, Steyger PS. Endotoxemia-mediated inflammation potentiates aminoglycoside-induced ototoxicity. Sci Transl Med. 2015;7(298):298ra118. doi: 10.1126/scitranslmed.aac5546. PubMed PMID: 26223301; PMCID: PMC4534720.
- Ahlstrom JB, Horwitz AR, Dubno JR. Spatial separation benefit for unaided and aided listening. Ear Hear. 2014;35(1):72–85. doi: 10.1097/AUD.0b013e3182a02274. PubMed PMID: 24121648; PMCID: PMC3872487.
- Roland JT, Jr., Gantz BJ, Waltzman SB, Parkinson AJ, Multicenter Clinical Trial G. United States multicenter clinical trial of the cochlear nucleus hybrid implant system. Laryngoscope. 2016;126(1):175–81. doi: 10.1002/lary.25451. PubMed PMID: 26152811; PMCID: PMC4704985.
- Quesnel AM, Nakajima HH, Rosowski JJ, Hansen MR, Gantz BJ, Nadol JB, Jr. Delayed loss of hearing after hearing preservation cochlear implantation: Human temporal bone pathology and implications for etiology. Hear Res. 2016;333:225–34. doi: 10.1016/j.heares.2015.08.018. PubMed PMID: 26341474; PMCID: PMC4775460.
- Reiss LA, Turner CW, Karsten SA, Gantz BJ. Plasticity in human pitch perception induced by tonotopically mismatched electro-acoustic stimulation. Neuroscience. 2014;256:43–52. doi: 10.1016/j.neuroscience.2013.10.024. PubMed PMID: 24157931; PMCID: PMC3893921.
- Friedmann DR, Ahmed OH, McMenomey SO, Shapiro WH, Waltzman SB, Roland JT, Jr. Single-sided Deafness Cochlear Implantation: Candidacy, Evaluation, and Outcomes in Children and Adults. Otol Neurotol. 2016;37(2):e154–60. doi: 10.1097/MAO.0000000000000951. PubMed PMID: 26756150.
- Mertens G, De Bodt M, Van de Heyning P. Cochlear implantation as a long-term treatment for ipsilateral incapacitating tinnitus in subjects with unilateral hearing loss up to 10 years. Hear Res. 2016;331:1–6. doi: 10.1016/j.heares.2015.09.016. PubMed PMID: 26433053.
- Bierer JA, Bierer SM, Kreft HA, Oxenham AJ. A fast method for measuring psychophysical thresholds across the cochlear implant array. Trends Hear. 2015;19. doi: 10.1177/2331216515569792. PubMed PMID: 25656797; PMCID: PMC4324086.
- van den Honert C, Kelsall DC. Focused intracochlear electric stimulation with phased array channels. J Acoust Soc Am. 2007;121(6):3703–16. doi: 10.1121/1.2722047. PubMed PMID: 17552721.
- Noij KS, Kozin ED, Sethi R, Shah PV, Kaplan AB, Herrmann B, Remenschneider A, Lee DJ. Systematic Review of Nontumor Pediatric Auditory Brainstem Implant Outcomes. Otolaryngol Head Neck Surg. 2015;153(5):739–50. doi: 10.1177/0194599815596929. PubMed PMID: 26227469.
- van de Berg R, Guinand N, Nguyen TA, Ranieri M, Cavuscens S, Guyot JP, Stokroos R, Kingma H, Perez-Fornos A. The vestibular implant: frequency-dependency of the electrically evoked vestibulo-ocular reflex in humans. Front Syst Neurosci. 2014;8:255. doi: 10.3389/fnsys.2014.00255. PubMed PMID: 25653601; PMCID: PMC4299437.
- Sun DQ, Lehar M, Dai C, Swarthout L, Lauer AM, Carey JP, Mitchell DE, Cullen KE, Della Santina CC. Histopathologic Changes of the Inner ear in Rhesus Monkeys After Intratympanic Gentamicin Injection and Vestibular Prosthesis Electrode Array Implantation. J Assoc Res Otolaryngol. 2015;16(3):373–87. doi: 10.1007/s10162–015–0515–y. PubMed PMID: 25790951; PMCID: PMC4417088.
- Phillips JO, Ling L, Nie K, Jameyson E, Phillips CM, Nowack AL, Golub JS, Rubinstein JT. Vestibular implantation and longitudinal electrical stimulation of the semicircular canal afferents in human subjects. J Neurophysiol. 2015;113(10):3866–92. doi: 10.1152/jn.00171.2013. PubMed PMID: 25652917; PMCID: PMC4480623.
- Auerbach BD, Rodrigues PV, Salvi RJ. Central gain control in tinnitus and hyperacusis. Front Neurol. 2014;5:206. doi: 10.3389/fneur.2014.00206. PubMed PMID: 25386157; PMCID: PMC4208401.
- Rauschecker JP, Leaver AM, Muhlau M. Tuning out the noise: limbic-auditory interactions in tinnitus. Neuron. 2010;66(6):819–26. doi: 10.1016/j.neuron.2010.04.032. PubMed PMID: 20620868; PMCID: PMC2904345.
- Hayes SH, Radziwon KE, Stolzberg DJ, Salvi RJ. Behavioral models of tinnitus and hyperacusis in animals. Front Neurol. 2014;5:179. doi: 10.3389/fneur.2014.00179. PubMed PMID: 25278931; PMCID: PMC4166233.
- Gu JW, Halpin CF, Nam EC, Levine RA, Melcher JR. Tinnitus, diminished sound-level tolerance, and elevated auditory activity in humans with clinically normal hearing sensitivity. J Neurophysiol. 2010;104(6):3361–70. doi: 10.1152/jn.00226.2010. PubMed PMID: 20881196; PMCID: PMC3007631.
- Chen YC, Li X, Liu L, Wang J, Lu CQ, Yang M, Jiao Y, Zang FC, Radziwon K, Chen GD, Sun W, Krishnan Muthaiah VP, Salvi R, Teng GJ. Tinnitus and hyperacusis involve hyperactivity and enhanced connectivity in auditory-limbic-arousal-cerebellar network. Elife. 2015;4:e06576. doi: 10.7554/eLife.06576. PubMed PMID: 25962854; PMCID: PMC4426664.
- Roberts LE, Eggermont JJ, Caspary DM, Shore SE, Melcher JR, Kaltenbach JA. Ringing ears: the neuroscience of tinnitus. J Neurosci. 2010;30(45):14972–9. doi: 10.1523/JNEUROSCI.4028–10.2010. PubMed PMID: 21068300; PMCID: PMC3073522.
- Piccirillo JF. Transcranial Magnetic Stimulation for Chronic Tinnitus. JAMA. 2016;315(5):506–7. doi: 10.1001/jama.2016.0075. PubMed PMID: 26836733.
- Shore SE, Roberts LE, Langguth B. Maladaptive plasticity in tinnitus - triggers, mechanisms and treatment. Nat Rev Neurol. 2016;12(3):150–60. doi: 10.1038/nrneurol.2016.12. PubMed PMID: 26868680; PMCID: PMC4895692.
- Hays SA, Rennaker RL, Kilgard MP. Targeting plasticity with vagus nerve stimulation to treat neurological disease. Prog Brain Res. 2013;207:275–99. doi: 10.1016/B978–0–444–63327–9.00010–2. PubMed PMID: 24309259; PMCID: PMC4615598.
- Pasley BN, David SV, Mesgarani N, Flinker A, Shamma SA, Crone NE, Knight RT, Chang EF. Reconstructing speech from human auditory cortex. PLoS Biol. 2012;10(1):e1001251. doi: 10.1371/journal.pbio.1001251. PubMed PMID: 22303281; PMCID: PMC3269422.
- Mesgarani N, Cheung C, Johnson K, Chang EF. Phonetic feature encoding in human superior temporal gyrus. Science. 2014;343(6174):1006–10. doi: 10.1126/science.1245994. PubMed PMID: 24482117; PMCID: PMC4350233.
- Bouchard KE, Mesgarani N, Johnson K, Chang EF. Functional organization of human sensorimotor cortex for speech articulation. Nature. 2013;495(7441):327–32. doi: 10.1038/nature11911. PubMed PMID: 23426266; PMCID: PMC3606666.
- Mesgarani N, David SV, Fritz JB, Shamma SA. Mechanisms of noise robust representation of speech in primary auditory cortex. Proc Natl Acad Sci U S A. 2014;111(18):6792–7. doi: 10.1073/pnas.1318017111. PubMed PMID: 24753585; PMCID: PMC4020083.
- Schneider DM, Woolley SM. Sparse and background-invariant coding of vocalizations in auditory scenes. Neuron. 2013;79(1):141–52. doi: 10.1016/j.neuron.2013.04.038. PubMed PMID: 23849201; PMCID: PMC3713513.
- Rabinowitz NC, Willmore BD, King AJ, Schnupp JW. Constructing noise-invariant representations of sound in the auditory pathway. PLoS Biol. 2013;11(11):e1001710. doi: 10.1371/journal.pbio.1001710. PubMed PMID: 24265596; PMCID: PMC3825667.
- Holt JC, Kewin K, Jordan PM, Cameron P, Klapczynski M, McIntosh JM, Crooks PA, Dwoskin LP, Lysakowski A. Pharmacologically distinct nicotinic acetylcholine receptors drive efferent-mediated excitation in calyx-bearing vestibular afferents. J Neurosci. 2015;35(8):3625–43. doi: 10.1523/JNEUROSCI.3388–14.2015. PubMed PMID: 25716861; PMCID: PMC4339364.
- Liu XP, Wooltorton JR, Gaboyard-Niay S, Yang FC, Lysakowski A, Eatock RA. Sodium channel diversity in the vestibular ganglion: NaV1.5, NaV1.8, and tetrodotoxin-sensitive currents. J Neurophysiol. 2016;115(5):2536–55. doi: 10.1152/jn.00902.2015. PubMed PMID: 26936982.
- Chen G, King JA, Burgess N, O'Keefe J. How vision and movement combine in the hippocampal place code. Proc Natl Acad Sci U S A. 2013;110(1):378–83. doi: 10.1073/pnas.1215834110. PubMed PMID: 23256159; PMCID: PMC3538268.
- Kaski D, Quadir S, Nigmatullina Y, Malhotra PA, Bronstein AM, Seemungal BM. Temporoparietal encoding of space and time during vestibular-guided orientation. Brain. 2016;139(Pt 2):392–403. doi: 10.1093/brain/awv370. PubMed PMID: 26719385; PMCID: PMC4805090.
- Fan RH, Liu S, DeAngelis GC, Angelaki DE. Heading Tuning in Macaque Area V6. J Neurosci. 2015;35(50):16303–14. doi: 10.1523/JNEUROSCI.2903–15.2015. PubMed PMID: 26674858; PMCID: PMC4679817.
- Dokka K, DeAngelis GC, Angelaki DE. Multisensory Integration of Visual and Vestibular Signals Improves Heading Discrimination in the Presence of a Moving Object. J Neurosci. 2015;35(40):13599–607. doi: 10.1523/JNEUROSCI.2267–15.2015. PubMed PMID: 26446214; PMCID: PMC4595618.
- Yau JM, DeAngelis GC, Angelaki DE. Dissecting neural circuits for multisensory integration and crossmodal processing. Philos Trans R Soc Lond B Biol Sci. 2015;370(1677):20140203. doi: 10.1098/rstb.2014.0203. PubMed PMID: 26240418; PMCID: PMC4528815.
- Carriot J, Jamali M, Cullen KE. Rapid adaptation of multisensory integration in vestibular pathways. Front Syst Neurosci. 2015;9:59. doi: 10.3389/fnsys.2015.00059. PubMed PMID: 25932009; PMCID: PMC4399207.
- CDC. Important Facts about Falls 2016. Available from: http://www.cdc.gov/homeandrecreationalsafety/falls/adultfalls.html.
- Formerly available from The NIH Senior Health website, discontinued as of 8/2017.
- NIH. Preventing Falls 2014; Winter 2014. Available from: https://www.nlm.nih.gov/medlineplus/magazine/issues/winter14/articles/winter14pg12-13.html.
- NIH. Dizziness Can Be a Drag: Coping with Balance Disorders 2012. Available from: https://newsinhealth.nih.gov/issue/aug2012/feature1.
- Orces CH, Alamgir H. Trends in fall-related injuries among older adults treated in emergency departments in the USA. Injury Prev. 2014;20(6):421–3. doi: 10.1136/injuryprev–2014–041268. PubMed PMID: WOS:000345276000009.
- CDC. National Center for Injury Prevention and Control. Web-based Injury Statistics Query and Reporting System (WISQARS) [online] [cited 2013 August 15].
- Buck L, Axel R. A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell. 1991;65(1):175–87. PubMed PMID: 1840504.
- NIDCD. NIDCD Taste and Smell Statistics 2016. Available from: https://www.nidcd.nih.gov/health/taste-smell.
- Rawal S, Hoffman HJ, Bainbridge KE, Huedo-Medina TB, Duffy VB. Prevalence and Risk Factors of Self-Reported Smell and Taste Alterations: Results from the 2011–2012 US National Health and Nutrition Examination Survey (NHANES). Chem Senses. 2016;41(1):69–76. doi: 10.1093/chemse/bjv057. PubMed PMID: 26487703; PMCID: PMC4715252.
- NIDDK. Overweight and Obesity Statistics2016.
- NIDDK. Do You Know Some of the Health Risks of Being Overweight? 2016.
- Beauchamp GK, Mennella JA. Flavor perception in human infants: development and functional significance. Digestion. 2011;83 Suppl 1:1–6. doi: 10.1159/000323397 PubMed PMID: 21389721; PMCID: PMC3202923.
- Dalton PH, Opiekun RE, Gould M, McDermott R, Wilson T, Maute C, Ozdener MH, Zhao K, Emmett E, Lees PS, Herbert R, Moline J. Chemosensory loss: functional consequences of the world trade center disaster. Environ Health Perspect. 2010;118(9):1251–6. doi: 10.1289/ehp.1001924. PubMed PMID: 20478761; PMCID: PMC2944085.
- Johnson MA, Tsai L, Roy DS, Valenzuela DH, Mosley C, Magklara A, Lomvardas S, Liberles SD, Barnea G. Neurons expressing trace amine-associated receptors project to discrete glomeruli and constitute an olfactory subsystem. Proc Natl Acad Sci U S A. 2012;109(33):13410–5. doi: 10.1073/pnas.1206724109. PubMed PMID: 22837392; PMCID: PMC3421222.
- Yee KK, Sukumaran SK, Kotha R, Gilbertson TA, Margolskee RF. Glucose transporters and ATP-gated K+ (KATP) metabolic sensors are present in type 1 taste receptor 3 (T1r3)-expressing taste cells. Proc Natl Acad Sci U S A. 2011;108(13):5431–6. doi: 10.1073/pnas.1100495108. PubMed PMID: 21383163; PMCID: PMC3069197.
- Yasumatsu K, Ogiwara Y, Takai S, Yoshida R, Iwatsuki K, Torii K, Margolskee RF, Ninomiya Y. Umami taste in mice uses multiple receptors and transduction pathways. J Physiol. 2012;590(5):1155–70. doi: 10.1113/jphysiol.2011.211920. PubMed PMID: 22183726; PMCID: PMC3381822.
- Ye W, Chang RB, Bushman JD, Tu YH, Mulhall EM, Wilson CE, Cooper AJ, Chick WS, Hill-Eubanks DC, Nelson MT, Kinnamon SC, Liman ER. The K+ channel KIR2.1 functions in tandem with proton influx to mediate sour taste transduction. Proc Natl Acad Sci U S A. 2016;113(2):E229–38. doi: 10.1073/pnas.1514282112. PubMed PMID: 26627720; PMCID: PMC4720319.
- Lewandowski BC, Sukumaran SK, Margolskee RF, Bachmanov AA. Amiloride-Insensitive Salt Taste Is Mediated by Two Populations of Type III Taste Cells with Distinct Transduction Mechanisms. J Neurosci. 2016;36(6):1942–53. doi: 10.1523/JNEUROSCI.2947–15.2016. PubMed PMID: 26865617; PMCID: PMC4748077.
- Taruno A, Vingtdeux V, Ohmoto M, Ma Z, Dvoryanchikov G, Li A, Adrien L, Zhao H, Leung S, Abernethy M, Koppel J, Davies P, Civan MM, Chaudhari N, Matsumoto I, Hellekant G, Tordoff MG, Marambaud P, Foskett JK. CALHM1 ion channel mediates purinergic neurotransmission of sweet, bitter and umami tastes. Nature. 2013;495(7440):223–6. doi: 10.1038/nature11906. PubMed PMID: 23467090; PMCID: PMC3600154.
- Chandrashekar J, Kuhn C, Oka Y, Yarmolinsky DA, Hummler E, Ryba NJ, Zuker CS. The cells and peripheral representation of sodium taste in mice. Nature. 2010;464(7286):297–301. doi: 10.1038/nature08783. PubMed PMID: 20107438; PMCID: PMC2849629.
- Li Q, Tachie-Baffour Y, Liu Z, Baldwin MW, Kruse AC, Liberles SD. Non-classical amine recognition evolved in a large clade of olfactory receptors. Elife. 2015;4:e10441. doi: 10.7554/eLife.10441. PubMed PMID: 26519734; PMCID: PMC4695389.
- Koh TW, He Z, Gorur-Shandilya S, Menuz K, Larter NK, Stewart S, Carlson JR. The Drosophila IR20a clade of ionotropic receptors are candidate taste and pheromone receptors. Neuron. 2014;83(4):850–65. doi: 10.1016/j.neuron.2014.07.012. PubMed PMID: 25123314; PMCID: PMC4141888.
- Hanchate NK, Kondoh K, Lu Z, Kuang D, Ye X, Qiu X, Pachter L, Trapnell C, Buck LB. Single-cell transcriptomics reveals receptor transformations during olfactory neurogenesis. Science. 2015;350(6265):1251–5. doi: 10.1126/science.aad2456. PubMed PMID: 26541607.
- Tizzano M, Gulbransen BD, Vandenbeuch A, Clapp TR, Herman JP, Sibhatu HM, Churchill ME, Silver WL, Kinnamon SC, Finger TE. Nasal chemosensory cells use bitter taste signaling to detect irritants and bacterial signals. Proc Natl Acad Sci U S A. 2010;107(7):3210–5. doi: 10.1073/pnas.0911934107. PubMed PMID: 20133764; PMCID: PMC2840287.
- Lee RJ, Kofonow JM, Rosen PL, Siebert AP, Chen B, Doghramji L, Xiong G, Adappa ND, Palmer JN, Kennedy DW, Kreindler JL, Margolskee RF, Cohen NA. Bitter and sweet taste receptors regulate human upper respiratory innate immunity. J Clin Invest. 2014;124(3):1393–405. doi: 10.1172/JCI72094. PubMed PMID: 24531552; PMCID: PMC3934184.
- McClintock TS, Adipietro K, Titlow WB, Breheny P, Walz A, Mombaerts P, Matsunami H. In vivo identification of eugenol-responsive and muscone-responsive mouse odorant receptors. J Neurosci. 2014;34(47):15669–78. doi: 10.1523/JNEUROSCI.3625–14.2014. PubMed PMID: 25411495; PMCID: PMC4236398.
- Munger SD, Leinders-Zufall T, McDougall LM, Cockerham RE, Schmid A, Wandernoth P, Wennemuth G, Biel M, Zufall F, Kelliher KR. An olfactory subsystem that detects carbon disulfide and mediates food-related social learning. Curr Biol. 2010;20(16):1438–44. doi: 10.1016/j.cub.2010.06.021. PubMed PMID: 20637621; PMCID: PMC2929674.
- Jiang Y, Gong NN, Hu XS, Ni MJ, Pasi R, Matsunami H. Molecular profiling of activated olfactory neurons identifies odorant receptors for odors in vivo. Nat Neurosci. 2015;18(10):1446–54. doi: 10.1038/nn.4104. PubMed PMID: 26322927; PMCID: PMC4583814.
- Cameron P, Hiroi M, Ngai J, Scott K. The molecular basis for water taste in Drosophila. Nature. 2010;465(7294):91–5. doi: 10.1038/nature09011. PubMed PMID: 20364123; PMCID: PMC2865571.
- Kaur AW, Ackels T, Kuo TH, Cichy A, Dey S, Hays C, Kateri M, Logan DW, Marton TF, Spehr M, Stowers L. Murine pheromone proteins constitute a context-dependent combinatorial code governing multiple social behaviors. Cell. 2014;157(3):676–88. doi: 10.1016/j.cell.2014.02.025. PubMed PMID: 24766811; PMCID: PMC4051225.
- Kass MD, Rosenthal MC, Pottackal J, McGann JP. Fear learning enhances neural responses to threat-predictive sensory stimuli. Science. 2013;342(6164):1389–92. doi: 10.1126/science.1244916. PubMed PMID: 24337299; PMCID: PMC4011636.
- Root CM, Ko KI, Jafari A, Wang JW. Presynaptic facilitation by neuropeptide signaling mediates odor-driven food search. Cell. 2011;145(1):133–44. doi: 10.1016/j.cell.2011.02.008. PubMed PMID: 21458672; PMCID: PMC3073827.
- Domingos AI, Vaynshteyn J, Voss HU, Ren X, Gradinaru V, Zang F, Deisseroth K, de Araujo IE, Friedman J. Leptin regulates the reward value of nutrient. Nat Neurosci. 2011;14(12):1562–8. doi: 10.1038/nn.2977. PubMed PMID: 22081158; PMCID: PMC4238286.
- Debiec J, Sullivan RM. Intergenerational transmission of emotional trauma through amygdala-dependent mother-to-infant transfer of specific fear. Proc Natl Acad Sci U S A. 2014;111(33):12222–7. doi: 10.1073/pnas.1316740111. PubMed PMID: 25071168; PMCID: PMC4142995.
- Yoshida R, Ohkuri T, Jyotaki M, Yasuo T, Horio N, Yasumatsu K, Sanematsu K, Shigemura N, Yamamoto T, Margolskee RF, Ninomiya Y. Endocannabinoids selectively enhance sweet taste. Proc Natl Acad Sci U S A. 2010;107(2):935–9. doi: 10.1073/pnas.0912048107. PubMed PMID: 20080779; PMCID: PMC2818929.
- Hayes JE, Sullivan BS, Duffy VB. Explaining variability in sodium intake through oral sensory phenotype, salt sensation and liking. Physiol Behav. 2010;100(4):369–80. doi: 10.1016/j.physbeh.2010.03.017. PubMed PMID: 20380843; PMCID: PMC2874635.
- Mathes CM, Bueter M, Smith KR, Lutz TA, le Roux CW, Spector AC. Roux-en-Y gastric bypass in rats increases sucrose taste-related motivated behavior independent of pharmacological GLP-1-receptor modulation. Am J Physiol Regul Integr Comp Physiol. 2012;302(6):R751–67. doi: 10.1152/ajpregu.00214.2011. PubMed PMID: 22170618; PMCID: PMC3774488.
- Pepino MY, Mennella JA. Habituation to the pleasure elicited by sweetness in lean and obese women. Appetite. 2012;58(3):800–5. doi: 10.1016/j.appet.2012.01.026. PubMed PMID: 22326885; PMCID: PMC3340467.
- Rudenga KJ, Small DM. Amygdala response to sucrose consumption is inversely related to artificial sweetener use. Appetite. 2012;58(2):504–7. doi: 10.1016/j.appet.2011.12.001. PubMed PMID: 22178008; PMCID: PMC3289048.
- Thiebaud N, Johnson MC, Butler JL, Bell GA, Ferguson KL, Fadool AR, Fadool JC, Gale AM, Gale DS, Fadool DA. Hyperlipidemic diet causes loss of olfactory sensory neurons, reduces olfactory discrimination, and disrupts odor-reversal learning. J Neurosci. 2014;34(20):6970–84. doi: 10.1523/JNEUROSCI.3366–13.2014. PubMed PMID: 24828650; PMCID: PMC4019806.
- Mennella JA, Finkbeiner S, Lipchock SV, Hwang LD, Reed DR. Preferences for salty and sweet tastes are elevated and related to each other during childhood. PLoS One. 2014;9(3):e92201. doi: 10.1371/journal.pone.0092201. PubMed PMID: 24637844; PMCID: PMC3956914.
- McIntyre JC, Davis EE, Joiner A, Williams CL, Tsai IC, Jenkins PM, McEwen DP, Zhang L, Escobado J, Thomas S, Szymanska K, Johnson CA, Beales PL, Green ED, Mullikin JC, Program NCS, Sabo A, Muzny DM, Gibbs RA, Attie-Bitach T, Yoder BK, Reed RR, Katsanis N, Martens JR. Gene therapy rescues cilia defects and restores olfactory function in a mammalian ciliopathy model. Nat Med. 2012;18(9):1423–8. doi: 10.1038/nm.2860. PubMed PMID: 22941275; PMCID: PMC3645984.
- Tadenev AL, Kulaga HM, May-Simera HL, Kelley MW, Katsanis N, Reed RR. Loss of Bardet-Biedl syndrome protein-8 (BBS8) perturbs olfactory function, protein localization, and axon targeting. Proc Natl Acad Sci U S A. 2011;108(25):10320–5. doi: 10.1073/pnas.1016531108. PubMed PMID: 21646512; PMCID: PMC3121838.
- Weiss J, Pyrski M, Jacobi E, Bufe B, Willnecker V, Schick B, Zizzari P, Gossage SJ, Greer CA, Leinders-Zufall T, Woods CG, Wood JN, Zufall F. Loss-of-function mutations in sodium channel Nav1.7 cause anosmia. Nature. 2011;472(7342):186–90. doi: 10.1038/nature09975. PubMed PMID: 21441906; PMCID: PMC3674497.
- Cramer PE, Cirrito JR, Wesson DW, Lee CY, Karlo JC, Zinn AE, Casali BT, Restivo JL, Goebel WD, James MJ, Brunden KR, Wilson DA, Landreth GE. ApoE-directed therapeutics rapidly clear beta-amyloid and reverse deficits in AD mouse models. Science. 2012;335(6075):1503–6. doi: 10.1126/science.1217697. PubMed PMID: 22323736; PMCID: PMC3651582.
- Cao L, Schrank BR, Rodriguez S, Benz EG, Moulia TW, Rickenbacher GT, Gomez AC, Levites Y, Edwards SR, Golde TE, Hyman BT, Barnea G, Albers MW. Abeta alters the connectivity of olfactory neurons in the absence of amyloid plaques in vivo. Nat Commun. 2012;3:1009. doi: 10.1038/ncomms2013. PubMed PMID: 22910355; PMCID: PMC3529477.
- Li W, Howard JD, Gottfried JA. Disruption of odour quality coding in piriform cortex mediates olfactory deficits in Alzheimer's disease. Brain. 2010;133(9):2714–26. doi: 10.1093/brain/awq209. PubMed PMID: 20724290; PMCID: PMC2948816.
- Lee RJ, Xiong G, Kofonow JM, Chen B, Lysenko A, Jiang P, Abraham V, Doghramji L, Adappa ND, Palmer JN, Kennedy DW, Beauchamp GK, Doulias PT, Ischiropoulos H, Kreindler JL, Reed DR, Cohen NA. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J Clin Invest. 2012;122(11):4145–59. doi: 10.1172/JCI64240. PubMed PMID: 23041624; PMCID: PMC3484455.
- Lane AP, Turner J, May L, Reed R. A genetic model of chronic rhinosinusitis-associated olfactory inflammation reveals reversible functional impairment and dramatic neuroepithelial reorganization. J Neurosci. 2010;30(6):2324–9. doi: 10.1523/JNEUROSCI.4507–09.2010. PubMed PMID: 20147558; PMCID: PMC2957830.
- Guo Z, Packard A, Krolewski RC, Harris MT, Manglapus GL, Schwob JE. Expression of pax6 and sox2 in adult olfactory epithelium. J Comp Neurol. 2010;518(21):4395–418. doi: 10.1002/cne.22463. PubMed PMID: 20852734; PMCID: PMC2940252.
- Wang YZ, Yamagami T, Gan Q, Wang Y, Zhao T, Hamad S, Lott P, Schnittke N, Schwob JE, Zhou CJ. Canonical Wnt signaling promotes the proliferation and neurogenesis of peripheral olfactory stem cells during postnatal development and adult regeneration. J Cell Sci. 2011;124(Pt 9):1553–63. doi: 10.1242/jcs.080580. PubMed PMID: 21486944; PMCID: PMC3078819.
- Yee KK, Li Y, Redding KM, Iwatsuki K, Margolskee RF, Jiang P. Lgr5-EGFP marks taste bud stem/progenitor cells in posterior tongue. Stem Cells. 2013;31(5):992–1000. doi: 10.1002/stem.1338. PubMed PMID: 23377989; PMCID: PMC3637415.
- Nguyen HM, Reyland ME, Barlow LA. Mechanisms of taste bud cell loss after head and neck irradiation. J Neurosci. 2012;32(10):3474–84. doi: 10.1523/JNEUROSCI.4167–11.2012. PubMed PMID: 22399770; PMCID: PMC3320161.
- Ren W, Lewandowski BC, Watson J, Aihara E, Iwatsuki K, Bachmanov AA, Margolskee RF, Jiang P. Single Lgr5- or Lgr6-expressing taste stem/progenitor cells generate taste bud cells ex vivo. Proc Natl Acad Sci U S A. 2014;111(46):16401–6. doi: 10.1073/pnas.1409064111. PubMed PMID: 25368147; PMCID: PMC4246268.
- Kumari A, Ermilov AN, Allen BL, Bradley RM, Dlugosz AA, Mistretta CM. Hedgehog pathway blockade with the cancer drug LDE225 disrupts taste organs and taste sensation. J Neurophysiol. 2015;113(3):1034–40. doi: 10.1152/jn.00822.2014. PubMed PMID: 25392175; PMCID: PMC4312875.
- Pellegrino M, Steinbach N, Stensmyr MC, Hansson BS, Vosshall LB. A natural polymorphism alters odour and DEET sensitivity in an insect odorant receptor. Nature. 2011;478(7370):511–4. doi: 10.1038/nature10438. PubMed PMID: 21937991; PMCID: PMC3203342.
- DeGennaro M, McBride CS, Seeholzer L, Nakagawa T, Dennis EJ, Goldman C, Jasinskiene N, James AA, Vosshall LB. orco mutant mosquitoes lose strong preference for humans and are not repelled by volatile DEET. Nature. 2013;498(7455):487–91. doi: 10.1038/nature12206. PubMed PMID: 23719379; PMCID: PMC3696029.
- Lee Y, Kim SH, Montell C. Avoiding DEET through insect gustatory receptors. Neuron. 2010;67(4):555–61. doi: 10.1016/j.neuron.2010.07.006. PubMed PMID: 20797533; PMCID: PMC2929391.
- Sosulski DL, Bloom ML, Cutforth T, Axel R, Datta SR. Distinct representations of olfactory information in different cortical centres. Nature. 2011;472(7342):213–6. doi: 10.1038/nature09868. PubMed PMID: 21451525; PMCID: PMC3354569.
- Chapuis J, Cohen Y, He X, Zhang Z, Jin S, Xu F, Wilson DA. Lateral entorhinal modulation of piriform cortical activity and fine odor discrimination. J Neurosci. 2013;33(33):13449–59. doi: 10.1523/JNEUROSCI.1387–13.2013. PubMed PMID: 23946403; PMCID: PMC3742931.
- Haddad R, Lanjuin A, Madisen L, Zeng H, Murthy VN, Uchida N. Olfactory cortical neurons read out a relative time code in the olfactory bulb. Nat Neurosci. 2013;16(7):949–57. doi: 10.1038/nn.3407. PubMed PMID: 23685720; PMCID: PMC3695490.
- Jezzini A, Mazzucato L, La Camera G, Fontanini A. Processing of hedonic and chemosensory features of taste in medial prefrontal and insular networks. J Neurosci. 2013;33(48):18966–78. doi: 10.1523/JNEUROSCI.2974–13.2013. PubMed PMID: 24285901; PMCID: PMC3841457.
- Miller P, Katz DB. Stochastic transitions between neural states in taste processing and decision-making. J Neurosci. 2010;30(7):2559–70. doi: 10.1523/JNEUROSCI.3047–09.2010. PubMed PMID: 20164341; PMCID: PMC2851230.
- Moran A, Katz DB. Sensory cortical population dynamics uniquely track behavior across learning and extinction. J Neurosci. 2014;34(4):1248–57. doi: 10.1523/JNEUROSCI.3331–13.2014. PubMed PMID: 24453316; PMCID: PMC3898286.
- Olofsson JK, Hurley RS, Bowman NE, Bao X, Mesulam MM, Gottfried JA. A designated odor-language integration system in the human brain. J Neurosci. 2014;34(45):14864–73. doi: 10.1523/JNEUROSCI.2247–14.2014. PubMed PMID: 25378154; PMCID: PMC4220022.
- Wesson DW, Borkowski AH, Landreth GE, Nixon RA, Levy E, Wilson DA. Sensory network dysfunction, behavioral impairments, and their reversibility in an Alzheimer's beta-amyloidosis mouse model. J Neurosci. 2011;31(44):15962–71. doi: 10.1523/JNEUROSCI.2085–11.2011. PubMed PMID: 22049439; PMCID: PMC3417321.
- Rokni D, Hemmelder V, Kapoor V, Murthy VN. An olfactory cocktail party: figure-ground segregation of odorants in rodents. Nat Neurosci. 2014;17(9):1225–32. doi: 10.1038/nn.3775. PubMed PMID: 25086608; PMCID: PMC4146660.
- Poo C, Isaacson JS. A major role for intracortical circuits in the strength and tuning of odor-evoked excitation in olfactory cortex. Neuron. 2011;72(1):41–8. doi: 10.1016/j.neuron.2011.08.015. PubMed PMID: 21982367; PMCID: PMC3190137.
- Kato HK, Chu MW, Isaacson JS, Komiyama T. Dynamic sensory representations in the olfactory bulb: modulation by wakefulness and experience. Neuron. 2012;76(5):962–75. doi: 10.1016/j.neuron.2012.09.037. PubMed PMID: 23217744; PMCID: PMC3523713.
- Doucette W, Gire DH, Whitesell J, Carmean V, Lucero MT, Restrepo D. Associative cortex features in the first olfactory brain relay station. Neuron. 2011;69(6):1176–87. doi: 10.1016/j.neuron.2011.02.024. PubMed PMID: 21435561; PMCID: PMC3064824.
- Bazhenov M, Huerta R, Smith BH. A computational framework for understanding decision making through integration of basic learning rules. J Neurosci. 2013;33(13):5686–97. doi: 10.1523/JNEUROSCI.4145–12.2013. PubMed PMID: 23536082; PMCID: PMC3667960.
- Harris DT, Kallman BR, Mullaney BC, Scott K. Representations of Taste Modality in the Drosophila Brain. Neuron. 2015;86(6):1449–60. doi: 10.1016/j.neuron.2015.05.026. PubMed PMID: 26051423; PMCID: PMC4474761.
- Wu A, Dvoryanchikov G, Pereira E, Chaudhari N, Roper SD. Breadth of tuning in taste afferent neurons varies with stimulus strength. Nat Commun. 2015;6:8171. doi: 10.1038/ncomms9171. PubMed PMID: 26373451; PMCID: PMC4573454.
- Wilson DM, Lemon CH. Modulation of central gustatory coding by temperature. J Neurophysiol. 2013;110(5):1117–29. doi: 10.1152/jn.00974.2012. PubMed PMID: 23761701; PMCID: PMC3763089.
- Barretto RP, Gillis-Smith S, Chandrashekar J, Yarmolinsky DA, Schnitzer MJ, Ryba NJ, Zuker CS. The neural representation of taste quality at the periphery. Nature. 2015;517(7534):373–6. doi: 10.1038/nature13873. PubMed PMID: 25383521; PMCID: PMC4297533.
- Wang W, Lu S, Li T, Pan YW, Zou J, Abel GM, Xu L, Storm DR, Xia Z. Inducible activation of ERK5 MAP kinase enhances adult neurogenesis in the olfactory bulb and improves olfactory function. J Neurosci. 2015;35(20):7833–49. doi: 10.1523/JNEUROSCI.3745–14.2015. PubMed PMID: 25995470; PMCID: PMC4438129.
- Howard JD, Gottfried JA. Configural and elemental coding of natural odor mixture components in the human brain. Neuron. 2014;84(4):857–69. doi: 10.1016/j.neuron.2014.10.012. PubMed PMID: 25453843; PMCID: PMC4254622.
- Fortis-Santiago Y, Rodwin BA, Neseliler S, Piette CE, Katz DB. State dependence of olfactory perception as a function of taste cortical inactivation. Nat Neurosci. 2010;13(2):158–9. doi: 10.1038/nn.2463. PubMed PMID: 20023656; PMCID: PMC2834247.
- Dus M, Lai JS, Gunapala KM, Min S, Tayler TD, Hergarden AC, Geraud E, Joseph CM, Suh GS. Nutrient Sensor in the Brain Directs the Action of the Brain-Gut Axis in Drosophila. Neuron. 2015;87(1):139–51. doi: 10.1016/j.neuron.2015.05.032. PubMed PMID: 26074004; PMCID: PMC4697866.
- Gaudry Q, Hong EJ, Kain J, de Bivort BL, Wilson RI. Asymmetric neurotransmitter release enables rapid odour lateralization in Drosophila. Nature. 2013;493(7432):424–8. doi: 10.1038/nature11747. PubMed PMID: 23263180; PMCID: PMC3590906.
- Gutierrez R, Simon SA, Nicolelis MA. Licking-induced synchrony in the taste-reward circuit improves cue discrimination during learning. J Neurosci. 2010;30(1):287–303. doi: 10.1523/JNEUROSCI.0855–09.2010. PubMed PMID: 20053910; PMCID: PMC2831544.
- Peng Y, Gillis-Smith S, Jin H, Trankner D, Ryba NJ, Zuker CS. Sweet and bitter taste in the brain of awake behaving animals. Nature. 2015;527(7579):512–5. doi: 10.1038/nature15763. PubMed PMID: 26580015; PMCID: PMC4712381.
- Root CM, Denny CA, Hen R, Axel R. The participation of cortical amygdala in innate, odour-driven behaviour. Nature. 2014;515(7526):269–73. doi: 10.1038/nature13897. PubMed PMID: 25383519; PMCID: PMC4231015.
- Dulac C, O'Connell LA, Wu Z. Neural control of maternal and paternal behaviors. Science. 2014;345(6198):765–70. doi: 10.1126/science.1253291. PubMed PMID: 25124430; PMCID: PMC4230532.
- van Breugel F, Riffell J, Fairhall A, Dickinson MH. Mosquitoes Use Vision to Associate Odor Plumes with Thermal Targets. Curr Biol. 2015;25(16):2123–9. doi: 10.1016/j.cub.2015.06.046. PubMed PMID: 26190071; PMCID: PMC4546539.
- McBride CS, Baier F, Omondi AB, Spitzer SA, Lutomiah J, Sang R, Ignell R, Vosshall LB. Evolution of mosquito preference for humans linked to an odorant receptor. Nature. 2014;515(7526):222–7. doi: 10.1038/nature13964. PubMed PMID: 25391959; PMCID: PMC4286346.
- CDC. Communication Disorders and Use of Intervention Services Among Children Aged 3–17 Years: United States, 2012 2012. Available from: http://www.cdc.gov/nchs/data/databriefs/db205.htm.
- Catts HW, Bridges MS, Little TD, Tomblin JB. Reading achievement growth in children with language impairments. J Speech Lang Hear Res. 2008;51(6):1569-79. doi: 10.1044/1092-4388(2008/07-0259). PubMed PMID: 18695010; PMCID: PMC3763805.
- Durkin K, Conti-Ramsden G. Language, social behavior, and the quality of friendships in adolescents with and without a history of specific language impairment. Child Dev. 2007;78(5):1441-57. doi: 10.1111/j.1467-8624.2007.01076.x. PubMed PMID: 17883441.
- Clegg J, Hollis C, Mawhood L, Rutter M. Developmental language disorders--a follow-up in later adult life. Cognitive, language and psychosocial outcomes. J Child Psychol Psychiatry. 2005;46(2):128-49. doi: 10.1111/j.1469-7610.2004.00342.x. PubMed PMID: 15679523.
- Rice ML, Haney KR, Wexler K. Family histories of children with SLI who show extended optional infinitives. J Speech Lang Hear Res. 1998;41(2):419–32. PubMed PMID: 9570593.
- Rice ML, Smith SD, Gayan J. Convergent genetic linkage and associations to language, speech and reading measures in families of probands with Specific Language Impairment. J Neurodev Disord. 2009;1(4):264–82. doi: 10.1007/s11689–009–9031–x. PubMed PMID: 19997522; PMCID: PMC2788915.
- Erickson E, Sivasankar M. Simulated reflux decreases vocal fold epithelial barrier resistance. Laryngoscope. 2010;120(8):1569–75. doi: 10.1002/lary.20983. PubMed PMID: 20564752; PMCID: PMC2927501.
- Levendoski EE, Sivasankar MP. Vocal fold ion transport and mucin expression following acrolein exposure. J Membr Biol. 2014;247(5):441–50. doi: 10.1007/s00232–014–9651–2. PubMed PMID: 24648011; PMCID: PMC4306594.
- Cohen SM, Kim J, Roy N, Asche C, Courey M. The impact of laryngeal disorders on work-related dysfunction. Laryngoscope. 2012;122(7):1589–94. doi: 10.1002/lary.23197. PubMed PMID: 22549455.
- Cohen SM, Kim J, Roy N, Asche C, Courey M. Direct health care costs of laryngeal diseases and disorders. Laryngoscope 2012;122(7):1582–8. doi: 10.1002/lary.23189. PubMed PMID: 22544473.
- Roy N, Merrill RM, Thibeault S, Parsa RA, Gray SD, Smith EM. Prevalence of voice disorders in teachers and the general population. J Speech Lang Hear Res. 2004;47(2):281–93. doi: 10.1044/1092–4388(2004/023). PubMed PMID: 15157130.
- Smith E, Lemke J, Taylor M, Kirchner HL, Hoffman H. Frequency of voice problems among teachers and other occupations. J Voice. 1998;12(4):480–8. PubMed PMID: 9988035.
- Thibeault SL, Merrill RM, Roy N, Gray SD, Smith EM. Occupational risk factors associated with voice disorders among teachers. Ann Epidemiol. 2004;14(10):786–92. doi: 10.1016/j.annepidem.2004.03.004. PubMed PMID: 15519901.
- Verdolini K, Ramig LO. Review: occupational risks for voice problems. Logoped Phoniatr Vocol. 2001;26(1):37–46. PubMed PMID: 11432413.
- Roger VL, Go AS, Lloyd-Jones DM, Adams RJ, Berry JD, Brown TM, Carnethon MR, Dai S, de Simone G, Ford ES, Fox CS, Fullerton HJ, Gillespie C, Greenlund KJ, Hailpern SM, Heit JA, Ho PM, Howard VJ, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Makuc DM, Marcus GM, Marelli A, Matchar DB, McDermott MM, Meigs JB, Moy CS, Mozaffarian D, Mussolino ME, Nichol G, Paynter NP, Rosamond WD, Sorlie PD, Stafford RS, Turan TN, Turner MB, Wong ND, Wylie-Rosett J, American Heart Association Statistics C, Stroke Statistics S. Heart disease and stroke statistics--2011 update: a report from the American Heart Association. Circulation. 2011;123(4):e18–e209. doi: 10.1161/CIR.0b013e3182009701. PubMed PMID: 21160056; PMCID: PMC4418670.
- Bays CL. Quality of life of stroke survivors: a research synthesis. J Neurosci Nurs. 2001;33(6):310–6. PubMed PMID: 11776713.
- Eicher JD, Powers NR, Miller LL, Mueller KL, Mascheretti S, Marino C, Willcutt EG, DeFries JC, Olson RK, Smith SD, Pennington BF, Tomblin JB, Ring SM, Gruen JR. Characterization of the DYX2 locus on chromosome 6p22 with reading disability, language impairment, and IQ. Hum Genet. 2014;133(7):869–81. doi: 10.1007/s00439–014–1427–3. PubMed PMID: 24509779; PMCID: PMC4053598.
- Fedorenko E, Morgan A, Murray E, Cardinaux A, Mei C, Tager-Flusberg H, Fisher SE, Kanwisher N. A highly penetrant form of childhood apraxia of speech due to deletion of 16p11.2. Eur J Hum Genet. 2016;24(2):302–6. doi: 10.1038/ejhg.2015.149. PubMed PMID: 26173965; PMCID: PMC4717199.
- Graham SA, Deriziotis P, Fisher SE. Insights into the genetic foundations of human communication. Neuropsychol Rev. 2015;25(1):3–26. doi: 10.1007/s11065–014–9277–2. PubMed PMID: 25597031.
- Kornilov SA, Rakhlin N, Koposov R, Lee M, Yrigollen C, Caglayan AO, Magnuson JS, Mane S, Chang JT, Grigorenko EL. Genome-Wide Association and Exome Sequencing Study of Language Disorder in an Isolated Population. Pediatrics. 2016;137(4). doi: 10.1542/peds.2015–2469. PubMed PMID: 27016271.
- Worthey EA, Raca G, Laffin JJ, Wilk BM, Harris JM, Jakielski KJ, Dimmock DP, Strand EA, Shriberg LD. Whole-exome sequencing supports genetic heterogeneity in childhood apraxia of speech. J Neurodev Disord. 2013;5(1):29. doi: 10.1186/1866–1955–5–29. PubMed PMID: 24083349; PMCID: PMC3851280.
- Lesca G, Rudolf G, Bruneau N, Lozovaya N, Labalme A, Boutry-Kryza N, Salmi M, Tsintsadze T, Addis L, Motte J, Wright S, Tsintsadze V, Michel A, Doummar D, Lascelles K, Strug L, Waters P, de Bellescize J, Vrielynck P, de Saint Martin A, Ville D, Ryvlin P, Arzimanoglou A, Hirsch E, Vincent A, Pal D, Burnashev N, Sanlaville D, Szepetowski P. GRIN2A mutations in acquired epileptic aphasia and related childhood focal epilepsies and encephalopathies with speech and language dysfunction. Nat Genet. 2013;45(9):1061–6. doi: 10.1038/ng.2726. PubMed PMID: 23933820.
- Turner SJ, Mayes AK, Verhoeven A, Mandelstam SA, Morgan AT, Scheffer IE. GRIN2A: an aptly named gene for speech dysfunction. Neurology. 2015;84(6):586–93. doi: 10.1212/WNL.0000000000001228. PubMed PMID: 25596506; PMCID: PMC4335991.
- Durkes A, Sivasankar MP. Bicarbonate availability for vocal fold epithelial defense to acidic challenge. Ann Otol Rhinol Laryngol. 2014;123(1):71–6. doi: 10.1177/0003489414521143. PubMed PMID: 24574427; PMCID: PMC4000062.
- Wood JM, Hussey DJ, Woods CM, Watson DI, Carney AS. Biomarkers and laryngopharyngeal reflux. J Laryngol Otol. 2011;125(12):1218–24. doi: 10.1017/S0022215111002234. PubMed PMID: 21914248.
- Welham NV, Ling C, Dawson JA, Kendziorski C, Thibeault SL, Yamashita M. Microarray-based characterization of differential gene expression during vocal fold wound healing in rats. Dis Model Mech. 2015;8(3):311–21. doi: 10.1242/dmm.018366. PubMed PMID: 25592437; PMCID: PMC4348567.
- Lewis BA, Patton E, Freebairn L, Tag J, Iyengar SK, Stein CM, Taylor HG. Psychosocial co-morbidities in adolescents and adults with histories of communication disorders. J Commun Disord. 2016;61:60–70. doi: 10.1016/j.jcomdis.2016.03.004. PubMed PMID: 27032038; PMCID: PMC4880534.
- Redmond SM. Language Impairment in the Attention-Deficit/Hyperactivity Disorder Context. J Speech Lang Hear Res. 2016;59(1):133–42. doi: 10.1044/2015_JSLHR–L–15–0038. PubMed PMID: 26502026; PMCID: PMC4867926.
- Snowling MJ, Duff FJ, Nash HM, Hulme C. Language profiles and literacy outcomes of children with resolving, emerging, or persisting language impairments. J Child Psychol Psychiatry. 2015. doi: 10.1111/jcpp.12497. PubMed PMID: 26681150.
- Stager SV, Freeman FJ, Braun A. Characteristics of Fluency and Speech in Two Families With High Incidences of Stuttering. J Speech Lang Hear Res. 2015;58(5):1440–51. doi: 10.1044/2015_JSLHR–S–14–0080. PubMed PMID: 26126023; PMCID: PMC4686306.
- Heris HK, Miri AK, Ghattamaneni NR, Li NY, Thibeault SL, Wiseman PW, Mongeau L. Microstructural and mechanical characterization of scarred vocal folds. J Biomech. 2015;48(4):708–11. doi: 10.1016/j.jbiomech.2015.01.014. PubMed PMID: 25648495; PMCID: PMC4337956.
- Kasari C, Brady N, Lord C, Tager-Flusberg H. Assessing the minimally verbal school-aged child with autism spectrum disorder. Autism Res. 2013;6(6):479–93. doi: 10.1002/aur.1334. PubMed PMID: 24353165; PMCID: PMC4139180.
- Szarkowski A, Mood D, Shield A, Wiley S, Yoshinaga-Itano C. A summary of current understanding regarding children with autism spectrum disorder who are deaf or hard of hearing. Semin Speech Lang. 2014;35(4):241–59. doi: 10.1055/s–0034–1389097. PubMed PMID: 25321849.
- Thurm A, Manwaring SS, Swineford L, Farmer C. Longitudinal study of symptom severity and language in minimally verbal children with autism. J Child Psychol Psychiatry. 2015;56(1):97–104. doi: 10.1111/jcpp.12285. PubMed PMID: 24961159; PMCID: PMC4581593.
- Varley R, Cowell PE, Dyson L, Inglis L, Roper A, Whiteside SP. Self-Administered Computer Therapy for Apraxia of Speech: Two-Period Randomized Control Trial With Crossover. Stroke. 2016;47(3):822–8. doi: 10.1161/STROKEAHA.115.011939. PubMed PMID: 26797664.
- Mehta DD, Van Stan JH, Zanartu M, Ghassemi M, Guttag JV, Espinoza VM, Cortes JP, Cheyne HA, 2nd, Hillman RE. Using Ambulatory Voice Monitoring to Investigate Common Voice Disorders: Research Update. Front Bioeng Biotechnol. 2015;3:155. doi: 10.3389/fbioe.2015.00155. PubMed PMID: 26528472; PMCID: PMC4607864.
- Mehta DD, Zanartu M, Feng SW, Cheyne HA, 2nd, Hillman RE. Mobile voice health monitoring using a wearable accelerometer sensor and a smartphone platform. IEEE Trans Biomed Eng. 2012;59(11):3090–6. doi: 10.1109/TBME.2012.2207896. PubMed PMID: 22875236; PMCID: PMC3539821.
- Kojima T, Valenzuela CV, Novaleski CK, Van Deusen M, Mitchell JR, Garrett CG, Sivasankar MP, Rousseau B. Effects of phonation time and magnitude dose on vocal fold epithelial genes, barrier integrity, and function. Laryngoscope. 2014;124(12):2770–8. doi: 10.1002/lary.24827. PubMed PMID: 25073715; PMCID: PMC4241156.
- Adams C, Lockton E, Freed J, Gaile J, Earl G, McBean K, Nash M, Green J, Vail A, Law J. The Social Communication Intervention Project: a randomized controlled trial of the effectiveness of speech and language therapy for school-age children who have pragmatic and social communication problems with or without autism spectrum disorder. Int J Lang Commun Disord. 2012;47(3):233–44. doi: 10.1111/j.1460–6984.2011.00146.x. PubMed PMID: 22512510.
- Kasari C, Kaiser A, Goods K, Nietfeld J, Mathy P, Landa R, Murphy S, Almirall D. Communication interventions for minimally verbal children with autism: a sequential multiple assignment randomized trial. J Am Acad Child Adolesc Psychiatry. 2014;53(6):635–46. doi: 10.1016/j.jaac.2014.01.019. PubMed PMID: 24839882; PMCID: PMC4030683.
- van Vuuren S, Cherney LR. A Virtual Therapist for Speech and Language Therapy. Intell Virtual Agents. 2014;8637:438–48. doi: 10.1007/978–3–319–09767–1_55. PubMed PMID: 25938137; PMCID: PMC4415876.
- Meinzer M, Darkow R, Lindenberg R, Floel A. Electrical stimulation of the motor cortex enhances treatment outcome in post-stroke aphasia. Brain. 2016;139(Pt 4):1152–63. doi: 10.1093/brain/aww002. PubMed PMID: 26912641.
- Tsapkini K, Frangakis C, Gomez Y, Davis C, Hillis AE. Augmentation of spelling therapy with transcranial direct current stimulation in primary progressive aphasia: Preliminary results and challenges. Aphasiology. 2014;28(8–9):1112–30. doi: 10.1080/02687038.2014.930410. PubMed PMID: 26097278; PMCID: PMC4470615.
- Engineer CT, Engineer ND, Riley JR, Seale JD, Kilgard MP. Pairing Speech Sounds With Vagus Nerve Stimulation Drives Stimulus-specific Cortical Plasticity. Brain Stimul. 2015;8(3):637–44. doi: 10.1016/j.brs.2015.01.408. PubMed PMID: 25732785; PMCID: PMC4461522.
- Boyd TK, Barnett JEH, More CM. Evaluating iPad Technology for Enhancing Communication Skills of Children With Autism Spectrum Disorders. Interv Sch Clin. 2015;51(1):19–27. doi: 10.1177/1053451215577476. PubMed PMID: WOS:000359419100003.
- McNaughton D, Light J. The iPad and mobile technology revolution: benefits and challenges for individuals who require augmentative and alternative communication. Augment Altern Commun. 2013;29(2):107–16. doi: 10.3109/07434618.2013.784930. PubMed PMID: 23705813.
- Guenther FH, Hickok G. Role of the auditory system in speech production. Handb Clin Neurol. 2015;129:161–75. doi: 10.1016/B978–0–444–62630–1.00009–3. PubMed PMID: 25726268.
- Fishman JM, Long J, Gugatschka M, De Coppi P, Hirano S, Hertegard S, Thibeault SL, Birchall MA. Stem cell approaches for vocal fold regeneration. Laryngoscope. 2016. doi: 10.1002/lary.25820. PubMed PMID: 26774977.
- Ling C, Li Q, Brown ME, Kishimoto Y, Toya Y, Devine EE, Choi KO, Nishimoto K, Norman IG, Tsegyal T, Jiang JJ, Burlingham WJ, Gunasekaran S, Smith LM, Frey BL, Welham NV. Bioengineered vocal fold mucosa for voice restoration. Sci Transl Med. 2015;7(314):314ra187. doi: 10.1126/scitranslmed.aab4014. PubMed PMID: 26582902; PMCID: PMC4669060.
- Bhattacharya P, Siegmund T. Computational modeling of vibration-induced systemic hydration of vocal folds over a range of phonation conditions. Int J Numer Method Biomed Eng. 2014;30(10):1019–43. doi: 10.1002/cnm.2642. PubMed PMID: 24760548.
- Ikuma T, Kunduk M, McWhorter AJ. Mitigation of temporal aliasing via harmonic modeling of laryngeal waveforms in high-speed videoendoscopy. J Acoust Soc Am. 2012;132(3):1636–45. doi: 10.1121/1.4742730. PubMed PMID: 22978892.
- Kvit AA, Devine EE, Jiang JJ, Vamos AC, Tao C. Characterizing liquid redistribution in a biphasic vibrating vocal fold using finite element analysis. J Voice. 2015;29(3):265–72. doi: 10.1016/j.jvoice.2014.08.010. PubMed PMID: 25619469; PMCID: PMC4439368.
- Samlan RA, Story BH, Lotto AJ, Bunton K. Acoustic and perceptual effects of left-right laryngeal asymmetries based on computational modeling. J Speech Lang Hear Res. 2014;57(5):1619–37. doi: 10.1044/2014_JSLHR–S–12–0405. PubMed PMID: 24845730; PMCID: PMC4495963.
- Yang A, Stingl M, Berry DA, Lohscheller J, Voigt D, Eysholdt U, Dollinger M. Computation of physiological human vocal fold parameters by mathematical optimization of a biomechanical model. J Acoust Soc Am. 2011;130(2):948–64. doi: 10.1121/1.3605551. PubMed PMID: 21877808; PMCID: PMC3195891.
- Port RG, Anwar AR, Ku M, Carlson GC, Siegel SJ, Roberts TP. Prospective MEG biomarkers in ASD: pre-clinical evidence and clinical promise of electrophysiological signatures. Yale J Biol Med. 2015;88(1):25–36. PubMed PMID: 25745372; PMCID: PMC4345535.
- Roberts TP, Heiken K, Zarnow D, Dell J, Nagae L, Blaskey L, Solot C, Levy SE, Berman JI, Edgar JC. Left hemisphere diffusivity of the arcuate fasciculus: influences of autism spectrum disorder and language impairment. AJNR Am J Neuroradiol. 2014;35(3):587–92. doi: 10.3174/ajnr.A3754. PubMed PMID: 24335547; PMCID: PMC3970288.
- Lu C, Qi Z, Harris A, Weil LW, Han M, Halverson K, Perrachione TK, Kjelgaard M, Wexler K, Tager-Flusberg H, Gabrieli JD. Shared neuroanatomical substrates of impaired phonological working memory across reading disability and autism. Biol Psychiatry Cogn Neurosci Neuroimaging. 2016;1(2):169–77. doi: 10.1016/j.bpsc.2015.11.001. PubMed PMID: 26949750; PMCID: PMC4776338.
- Fuertinger S, Horwitz B, Simonyan K. The Functional Connectome of Speech Control. PLoS Biol. 2015;13(7):e1002209. doi: 10.1371/journal.pbio.1002209. PubMed PMID: 26204475; PMCID: PMC4512708.
- Behroozmand R, Shebek R, Hansen DR, Oya H, Robin DA, Howard MA, 3rd, Greenlee JD. Sensory-motor networks involved in speech production and motor control: an fMRI study. Neuroimage. 2015;109:418–28. doi: 10.1016/j.neuroimage.2015.01.040. PubMed PMID: 25623499; PMCID: PMC4339397.
- Lametti DR, Rochet-Capellan A, Neufeld E, Shiller DM, Ostry DJ. Plasticity in the human speech motor system drives changes in speech perception. J Neurosci. 2014;34(31):10339–46. doi: 10.1523/JNEUROSCI.0108–14.2014. PubMed PMID: 25080594; PMCID: PMC4115140.
- Simmonds AJ, Leech R, Collins C, Redjep O, Wise RJ. Sensory-motor integration during speech production localizes to both left and right plana temporale. J Neurosci. 2014;34(39):12963–72. doi: 10.1523/JNEUROSCI.0336–14.2014. PubMed PMID: 25253845; PMCID: PMC4172797.
- Simonyan K, Fuertinger S. Speech networks at rest and in action: interactions between functional brain networks controlling speech production. J Neurophysiol. 2015;113(7):2967–78. doi: 10.1152/jn.00964.2014. PubMed PMID: 25673742; PMCID: PMC4416556.
- Correia JM, Jansma BM, Bonte M. Decoding Articulatory Features from fMRI Responses in Dorsal Speech Regions. J Neurosci. 2015;35(45):15015–25. doi: 10.1523/JNEUROSCI.0977–15.2015. PubMed PMID: 26558773.
- Humphries C, Sabri M, Lewis K, Liebenthal E. Hierarchical organization of speech perception in human auditory cortex. Front Neurosci. 2014;8:406. doi: 10.3389/fnins.2014.00406. PubMed PMID: 25565939; PMCID: PMC4263085.
- Hurschler MA, Liem F, Oechslin M, Stampfli P, Meyer M. fMRI reveals lateralized pattern of brain activity modulated by the metrics of stimuli during auditory rhyme processing. Brain Lang. 2015;147:41–50. doi: 10.1016/j.bandl.2015.05.004. PubMed PMID: 26025759.
- Leaver AM, Rauschecker JP. Functional Topography of Human Auditory Cortex. J Neurosci. 2016;36(4):1416–28. doi: 10.1523/JNEUROSCI.0226–15.2016. PubMed PMID: 26818527; PMCID: PMC4728734.
- Mesgarani N, Chang EF. Selective cortical representation of attended speaker in multi-talker speech perception. Nature. 2012;485(7397):233–6. doi: 10.1038/nature11020. PubMed PMID: 22522927; PMCID: PMC3870007.
- Nourski KV, Brugge JF, Reale RA, Kovach CK, Oya H, Kawasaki H, Jenison RL, Howard MA, 3rd. Coding of repetitive transients by auditory cortex on posterolateral superior temporal gyrus in humans: an intracranial electrophysiology study. J Neurophysiol. 2013;109(5):1283–95. doi: 10.1152/jn.00718.2012. PubMed PMID: 23236002; PMCID: PMC3602837.
- Nourski KV, Steinschneider M, McMurray B, Kovach CK, Oya H, Kawasaki H, Howard MA, 3rd. Functional organization of human auditory cortex: investigation of response latencies through direct recordings. Neuroimage. 2014;101:598–609. doi: 10.1016/j.neuroimage.2014.07.004. PubMed PMID: 25019680; PMCID: PMC4430832.
- Sedley W, Gander PE, Kumar S, Kovach CK, Oya H, Kawasaki H, Howard MA, Griffiths TD. Neural signatures of perceptual inference. Elife. 2016;5. doi: 10.7554/eLife.11476. PubMed PMID: 26949254; PMCID: PMC4841773.
- Sammler D, Grosbras MH, Anwander A, Bestelmeyer PE, Belin P. Dorsal and Ventral Pathways for Prosody. Curr Biol. 2015;25(23):3079–85. doi: 10.1016/j.cub.2015.10.009. PubMed PMID: 26549262.
- Weisberg J, McCullough S, Emmorey K. Simultaneous perception of a spoken and a signed language: The brain basis of ASL-English code-blends. Brain Lang. 2015;147:96–106. doi: 10.1016/j.bandl.2015.05.006. PubMed PMID: 26177161.
- Beal DS, Gracco VL, Brettschneider J, Kroll RM, De Nil LF. A voxel-based morphometry (VBM) analysis of regional grey and white matter volume abnormalities within the speech production network of children who stutter. Cortex. 2013;49(8):2151–61. doi: 10.1016/j.cortex.2012.08.013. PubMed PMID: 23140891; PMCID: PMC3617061.
- Chang SE, Zhu DC. Neural network connectivity differences in children who stutter. Brain. 2013;136(Pt 12):3709–26. doi: 10.1093/brain/awt275. PubMed PMID: 24131593; PMCID: PMC3859219.
- Chang SE, Zhu DC, Choo AL, Angstadt M. White matter neuroanatomical differences in young children who stutter. Brain. 2015;138(Pt 3):694–711. doi: 10.1093/brain/awu400. PubMed PMID: 25619509; PMCID: PMC4339778.
- Neef NE, Anwander A, Friederici AD. The Neurobiological Grounding of Persistent Stuttering: from Structure to Function. Curr Neurol Neurosci Rep. 2015;15(9):63. doi: 10.1007/s11910–015–0579–4. PubMed PMID: 26228377.
- Kirke DN, Battistella G, Kumar V, Rubien-Thomas E, Choy M, Rumbach A, Simonyan K. Neural correlates of dystonic tremor: a multimodal study of voice tremor in spasmodic dysphonia. Brain Imaging Behav. 2016. doi: 10.1007/s11682–016–9513–x. PubMed PMID: 26843004.
- Simonyan K, Berman BD, Herscovitch P, Hallett M. Abnormal striatal dopaminergic neurotransmission during rest and task production in spasmodic dysphonia. J Neurosci. 2013;33(37):14705–14. doi: 10.1523/JNEUROSCI.0407–13.2013. PubMed PMID: 24027271; PMCID: PMC3771037.
- Termsarasab P, Ramdhani RA, Battistella G, Rubien-Thomas E, Choy M, Farwell IM, Velickovic M, Blitzer A, Frucht SJ, Reilly RB, Hutchinson M, Ozelius LJ, Simonyan K. Neural correlates of abnormal sensory discrimination in laryngeal dystonia. Neuroimage Clin. 2016;10:18–26. doi: 10.1016/j.nicl.2015.10.016. PubMed PMID: 26693398; PMCID: PMC4660380.
- Mesulam MM, Weintraub S, Rogalski EJ, Wieneke C, Geula C, Bigio EH. Asymmetry and heterogeneity of Alzheimer's and frontotemporal pathology in primary progressive aphasia. Brain. 2014;137(Pt 4):1176–92. doi: 10.1093/brain/awu024. PubMed PMID: 24574501; PMCID: PMC3959558.
- Mirman D, Chen Q, Zhang Y, Wang Z, Faseyitan OK, Coslett HB, Schwartz MF. Neural organization of spoken language revealed by lesion-symptom mapping. Nat Commun. 2015;6:6762. doi: 10.1038/ncomms7762. PubMed PMID: 25879574; PMCID: PMC4400840.
- Kiran S, Meier EL, Kapse KJ, Glynn PA. Changes in task-based effective connectivity in language networks following rehabilitation in post-stroke patients with aphasia. Front Hum Neurosci. 2015;9:316. doi: 10.3389/fnhum.2015.00316. PubMed PMID: 26106314; PMCID: PMC4460429.
- Tsui PH, Wan YL, Chen CK. Ultrasound imaging of the larynx and vocal folds: recent applications and developments. Curr Opin Otolaryngol Head Neck Surg. 2012;20(6):437–42. doi: 10.1097/MOO.0b013e32835896b4. PubMed PMID: 23000732.
- Lewis BA, Freebairn L, Tag J, Ciesla AA, Iyengar SK, Stein CM, Taylor HG. Adolescent outcomes of children with early speech sound disorders with and without language impairment. Am J Speech Lang Pathol. 2015;24(2):150–63. doi: 10.1044/2014_AJSLP–14–0075. PubMed PMID: 25569242; PMCID: PMC4477798.
- Rice ML, Hoffman L. Predicting vocabulary growth in children with and without specific language impairment: a longitudinal study from 2;6 to 21 years of age. J Speech Lang Hear Res. 2015;58(2):345–59. doi: 10.1044/2015_JSLHR–L–14–0150. PubMed PMID: 25611623; PMCID: PMC4398600.
- Ellis Weismer S, Kover ST. Preschool language variation, growth, and predictors in children on the autism spectrum. J Child Psychol Psychiatry. 2015;56(12):1327–37. doi: 10.1111/jcpp.12406. PubMed PMID: 25753577; PMCID: PMC4565784.
- Tager-Flusberg H. Risk Factors Associated With Language in Autism Spectrum Disorder: Clues to Underlying Mechanisms. J Speech Lang Hear Res. 2016;59(1):143–54. doi: 10.1044/2015_JSLHR–L–15–0146. PubMed PMID: 26502110; PMCID: PMC4867927.
- Woynaroski T, Watson L, Gardner E, Newsom CR, Keceli-Kaysili B, Yoder PJ. Early Predictors of Growth in Diversity of Key Consonants Used in Communication in Initially Preverbal Children with Autism Spectrum Disorder. J Autism Dev Disord. 2016;46(3):1013–24. doi: 10.1007/s10803–015–2647–7. PubMed PMID: 26603885; PMCID: PMC4747804.
- Elsabbagh M, Hohenberger A, Campos R, Van Herwegen J, Serres J, de Schonen S, Aschersleben G, Karmiloff-Smith A. Narrowing perceptual sensitivity to the native language in infancy: exogenous influences on developmental timing. Behav Sci (Basel). 2013;3(1):120–32. doi: 10.3390/bs3010120. PubMed PMID: 25379229; PMCID: PMC4217615.
- Berken JA, Chai X, Chen JK, Gracco VL, Klein D. Effects of Early and Late Bilingualism on Resting-State Functional Connectivity. J Neurosci. 2016;36(4):1165–72. doi: 10.1523/JNEUROSCI.1960–15.2016. PubMed PMID: 26818505.
- Krizman J, Tierney A, Fitzroy AB, Skoe E, Amar J, Kraus N. Continued maturation of auditory brainstem function during adolescence: A longitudinal approach. Clin Neurophysiol. 2015;126(12):2348–55. doi: 10.1016/j.clinph.2015.01.026. PubMed PMID: 25801342; PMCID: PMC4550576.
- Perez CA, Engineer CT, Jakkamsetti V, Carraway RS, Perry MS, Kilgard MP. Different timescales for the neural coding of consonant and vowel sounds. Cereb Cortex. 2013;23(3):670–83. doi: 10.1093/cercor/bhs045. PubMed PMID: 22426334; PMCID: PMC3563339.
- Lungova V, Verheyden JM, Herriges J, Sun X, Thibeault SL. Ontogeny of the mouse vocal fold epithelium. Dev Biol. 2015;399(2):263–82. doi: 10.1016/j.ydbio.2014.12.037. PubMed PMID: 25601450; PMCID: PMC4352410.
- Patel R, Donohue KD, Unnikrishnan H, Kryscio RJ. Kinematic measurements of the vocal-fold displacement waveform in typical children and adult populations: quantification of high-speed endoscopic videos. J Speech Lang Hear Res. 2015;58(2):227–40. doi: 10.1044/2015_JSLHR–S–13–0056. PubMed PMID: 25652615; PMCID: PMC4675116.
- Patel R, Dubrovskiy D, Dollinger M. Characterizing vibratory kinematics in children and adults with high-speed digital imaging. J Speech Lang Hear Res. 2014;57(2):S674–86. doi: 10.1044/2014_JSLHR–S–12–0278. PubMed PMID: 24686982.
- Branco A, Rodrigues SA, Fabro AT, Fonseca-Alves CE, Martins RH. Hyaluronic acid behavior in the lamina propria of the larynx with advancing age. Otolaryngol Head Neck Surg. 2014;151(4):652–6. doi: 10.1177/0194599814544673. PubMed PMID: 25096358.
- Branco A, Todorovic Fabro A, Goncalves TM, Garcia Martins RH. Alterations in extracellular matrix composition in the aging larynx. Otolaryngol Head Neck Surg. 2015;152(2):302–7. doi: 10.1177/0194599814562727. PubMed PMID: 25645525.
- Martins RH, Benito Pessin AB, Nassib DJ, Branco A, Rodrigues SA, Matheus SM. Aging voice and the laryngeal muscle atrophy. Laryngoscope. 2015;125(11):2518–21. doi: 10.1002/lary.25398. PubMed PMID: 26154530.
- Moore J, Thibeault S. Insights into the role of elastin in vocal fold health and disease. J Voice. 2012;26(3):269–75. doi: 10.1016/j.jvoice.2011.05.003. PubMed PMID: 21708449; PMCID: PMC3190022.
- Roberts T, Morton R, Al-Ali S. Microstructure of the vocal fold in elderly humans. Clin Anat. 2011;24(5):544–51. doi: 10.1002/ca.21114. PubMed PMID: 21647958.
- NIDCD. Spasmodic Dysphonia Fact Sheet 2010. Available from: /health/spasmodic-dysphonia.

