A new agreement announced January 7 between researchers at the National Institutes of Health (NIH) and a biotechnology company provides funding to continue an important clinical treatment study for Niemann-Pick disease type C (NPC), a debilitating and often fatal genetic disease. Audiologists and other research staff from the NIH’s National Institute on Deafness and Other Communication Disorders (NIDCD) will continue to play a key role in the ongoing research being conducted at the NIH’s Clinical Center. Scientists from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) are leading the phase I clinical trial of cyclodextrin, a promising new therapy for NPC.
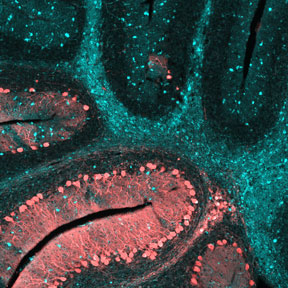
This image shows the cerebellum of a brain
affected by NPC at the end stage of the disease.
The blue staining shows the dense pockets of
lipid accumulations throughout the brain.
Funding from Vtesse, Inc., a company based in Gaithersburg, Maryland, will also support new clinical and laboratory studies on potential treatments for NPC and other lysosomal storage disorders, to be conducted in collaboration with NICHD, the National Center for Advancing Translational Sciences (NCATS), and other NIH components.
Also known as lipid storage diseases, lysosomal storage disorders primarily affect children, and some, such as NPC, may be fatal by early adulthood. Fatty materials in the cells and tissues of the body accumulate, damaging the brain, peripheral nervous system, liver, and other organs and tissues. Patients with NPC develop problems such as trouble swallowing and walking, and may progressively lose their vision and hearing.
There is currently no effective treatment for NPC. Researchers in National Center for Advancing Translational Sciences’ Therapeutics for Rare and Neglected Diseases (TRND) program developed cyclodextrin as part of a project focused on finding treatments for NPC. The goal of TRND projects is to develop therapeutic approaches in the laboratory to the stage where they attract biotechnology and pharmaceutical companies to invest in their further clinical development. The NPC project is a collaboration among government, academic, and industry researchers, who worked with patient groups and the NPC community to advance knowledge about the disease.
Ongoing studies by the NIDCD have demonstrated that NPC contributes to significant and progressive hearing loss. Confounding this is evidence that cyclodextrin has been shown to contribute to hearing loss by damaging the ear. During the phase I clinical trial, NIDCD research audiologists are closely monitoring the hearing of study volunteers to determine what doses of cyclodextrin are dangerous for hearing and who may be most at risk for hearing loss. Together with NIDCD otologist, Michael Hoa, M.D., the audiology team plays an active role in supporting families enrolled in the trial and helping those with hearing loss to obtain appropriate management and care.
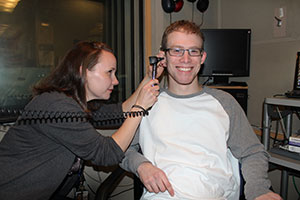
NIDCD research audiologist Kelly King, Au.D., Ph.D.,
examines Chris Fortin’s hearing at the Audiology Unit
at the NIH Clinical Center. Fortin is a 22-year-old
volunteer in the clinical trial to test the safety of a new
drug for Niemann-Pick disease type C, a rare and
debilitating disorder that is often fatal by early adulthood.
Through ongoing collaboration with other NIH scientists, researchers within the NIDCD Audiology Unit have contributed significantly to scientists’ understanding of the natural history of NPC as it relates to hearing function. Since 2006, the NIDCD research team has evaluated the hearing of more than 80 patients with NPC—the largest and most comprehensive collection of hearing data in individuals with this debilitating condition. In early 2014, the researchers reported that nearly 75 percent of the NPC patients studied have functionally significant hearing loss as a result of their disease. The participants ranged in age from four months to 21 years, with an average age of 9 years.
NIDCD researchers have also conducted studies in mice with NPC to better understand the role of cholesterol in the auditory system and the mechanisms involved in hearing loss linked to NPC. Collectively, their work has led to important recommendations on the care and management of patients with NPC.
“People with NPC should have their hearing monitored regularly by an audiologist so that care providers and family members can know when hearing loss is beginning, and so they can take steps to help the patient adjust with assistive devices, such as hearing aids, when appropriate,” notes Kelly King, Au.D., Ph.D., NIDCD research audiologist. “In many cases, these patients are unable to self-report changes in their hearing because of their young age or other neurological effects of the condition, so regular monitoring is key. In our natural history study, 40 percent of participants or their families were unaware that the child had hearing loss.”
The current study is one of dozens of research protocols for which the NIDCD Audiology Unit provides hearing exams and collects and assesses valuable information on hearing function in research volunteers to study the natural history of disorders that affect the auditory and vestibular (balance) systems, monitor the impact of treatments and diagnostic tests on hearing function, and study auditory processing skills.
For more information:
- NIH teams with industry to develop treatments for Niemann-Pick disease Type C, NIH news release, January 7, 2015
- NIDCD Audiology Unit
- King KA, et al. (2014) Auditory phenotype of Niemann-Pick disease, type C1. Ear and Hearing 35(1):110-117.
- King KA, et al. (2014) Hearing loss is an early consequence of Npc1 gene deletion in the mouse model of Niemann-Pick disease, type C. Journal of the Association for Research in Otolaryngology (JARO) 15(4):529-541.

