Drug development, from discovery to clinic, can be extremely costly. The NIDCD annual budget does not allow support for all stages of drug development. The institute does, however, encourage and support drug development to the greatest extent possible, and this includes leveraging existing federal and private resources as well as engaging in collaborative activities.
Investigators interested in support for drug development may wish to explore the programs listed below. Each has specific guidelines and project requirements so read the announcements thoroughly and contact the listed staff with any questions. While these funding opportunities do not support comprehensive coverage through the discovery and development pipeline, they provide funding options that may advance projects to the next stage and toward further funding opportunities.
The simplified schematic below provides an example of where NIH programs might contribute during the small molecule pipeline—from discovery to development. Entry and exit points and the therapeutic specificity of any given announcement vary depending on the funding program.
Discovery
- NIH Standard R01: NIH Research Project Grant (Parent R01 Clinical Trial Not Allowed)
- Exploratory R21: NIH Exploratory/Developmental Research Grant Program (Parent R21 Clinical Trial Not Allowed)
- NIDCD Translational R01: NIDCD Research Grants for Translating Basic Research into Clinical Tools (R01)
- SBIR/STTR: SBIR/STTR Funding
- NIDCD P50: Clinical Research Center Grant (P50 Clinical Trial Optional)
- Numerous Trans-NIH program announcements (PAs/PARs):
- Engineering Next-Generation Human Nervous System Microphysiological Systems (R01 Clinical Trials Not Allowed)
- Engineering Next-Generation Human Nervous System Microphysiological Systems (R21 Clinical Trials Not Allowed)
- Innovation Grants to Nurture Initial Translational Efforts (IGNITE): Neurotherapeutic Agent Characterization and In vivo Efficacy Studies (R61/R33 Clinical Trial Not Allowed)
Development and Beyond
-
- NCATS CTSA: Clinical and Translational Science Awards Program Projects & Initiatives
- NCATS Drug Development Collaboratory: Drug Development Collaboratory (UG3/UH3 Clinical Trial Required) (PDF | 1.5MB)
- NINDS Blueprint Neurotherapeutics: Blueprint Neurotherapeutics Network (BPN) for Small Molecules
- NCATS BrIDGs: Bridging Interventional Development Gaps (BrIDGs)
- NCATS TRND: Therapeutics for Rare and Neglected Diseases (TRND)
- NCI NExT: NCI Experimental Therapeutics Program
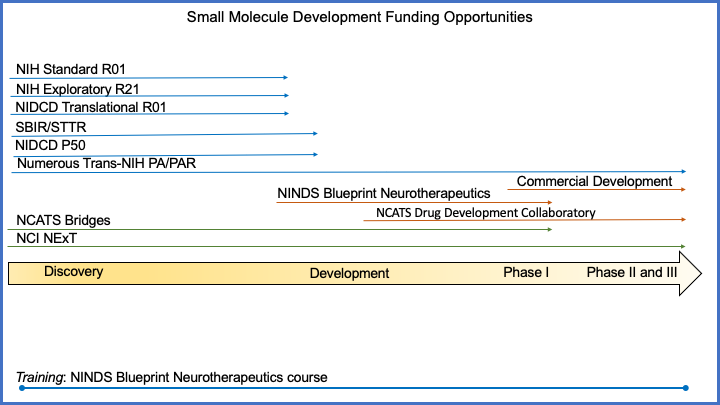
A National Institute of Neurological Disorders and Stroke (NINDS)-supported course on the complexity of drug discovery and development provides a training opportunity for those interested in this topic.
*Note: PDF files can be viewed with the free Adobe Reader.

