- Download Congressional Justification 2020 (855 KB PDF)
FY 2020 Budget
DEPARTMENT OF HEALTH AND HUMAN SERVICES
NATIONAL INSTITUTES OF HEALTH
National Institute on Deafness and Other Communication Disorders (NIDCD)
On this page:
- Organization Chart
- Appropriation Language
- Amounts Available for Obligation
- Budget Mechanism Table
- Major Changes in Budget Request
- Summary of Changes
- Budget Graphs
- Budget Authority by Activity
- Authorizing Legislation
- Appropriations History
- Justification of Budget Request
- Budget Authority by Object Class
- Salaries and Expenses
- Detail of Full-Time Equivalent Employment (FTE)
- Detail of Positions
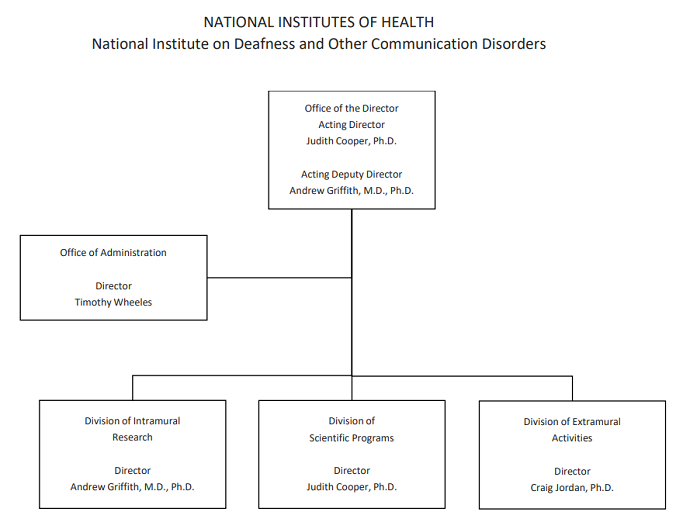
Organization Chart
Appropriation Language
NATIONAL INSTITUTES OF HEALTH
National Institute on Deafness and Other Communication Disorders
For carrying out section 301 and title IV of the PHS Act with respect to deafness and other communication disorders, [$474,404,000]$408,358,000.
| (Dollars in Thousands) | |||
|---|---|---|---|
| Source of Funding | FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget |
| 1 Excludes the following amounts (in thousands) for reimbursable activities carried out by this account: FY 2018 - $1,487; FY 2019 - $2,500; FY 2020 - $2,500 | |||
| Appropriation | $459, 974 | $474, 404 | $408, 358 |
| Mandatory Appropriation: (non-add) | |||
| Type 1 Diabetes | (0) | (0) | (0) |
| Other Mandatory financing | (0) | (0) | (0) |
| Rescission | 0 | 0 | 0 |
| Sequestration | 0 | 0 | 0 |
| Secretary's Transfer | -1,081 | 0 | 0 |
| OAR HIV/AIDS Transfers | 0 | 0 | 0 |
| Subtotal, adjusted budget authority | $458,893 | $474,404 | $408,358 |
| Unobligated balance, end of year | 0 | 0 | 0 |
| Subtotal, adjusted budget authority | $458,893 | $474,404 | $408,358 |
| Unobligated balance lapsing | -17 | 0 | 0 |
| Total obligations | $458,876 | $474,404 | $408,358 |
| (Dollars in Thousands) | ||||||||
|---|---|---|---|---|---|---|---|---|
| MECHANISM | FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY2019 CR | ||||
| No. | Amount | No. | Amount | No. | Amount | No. | Amount | |
| 1All items in italics and brackets are non-add entries. | ||||||||
| Research Projects: | ||||||||
| Noncompeting | 586 | $231,567 | 624 | $251,469 | 643 | $243,990 | 19 | -$7,479 |
| Administrative Supplements | (38) | 2,954 | (30) | 2,200 | (15) | 1,000 | (-15) | -1,200 |
| Competing | ||||||||
| Renewal | 49 | 24,278 | 41 | 20.364 | 22 | 9,834 | -19 | -10,530 |
| New | 179 | 68,908 | 150 | 57,984 | 76 | 26,441 | -74 | -31,543 |
| Supplements | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Subtotal, Competing | 228 | $93, 186 | 191 | $78,348 | 98 | $36,275 | -93 | -$42,073 |
| Subtotal, RPGs | 814 | $327,707 | 815 | $332,017 | 741 | $281,265 | -74 | -$50,752 |
| SBIR/STTR | 36 | 14,423 | 37 | 14,915 | 33 | 12,842 | -4 | -2,073 |
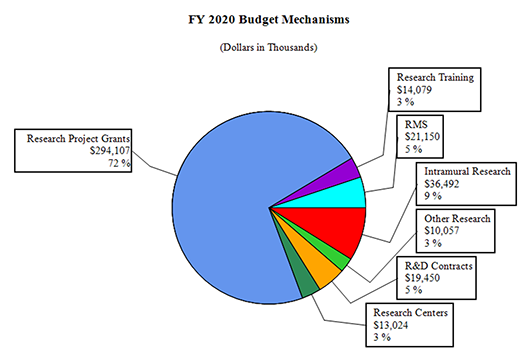
| Research Project Grants | 850 | $342,130 | 852 | $346,932 | 774 | $294,107 | -78 | -$52,825 |
| Research Centers: | ||||||||
| Specialized/Comprehensive | 5 | $11,652 | 6 | $14,471 | 6 | $13,024 | 0 | -$1,447 |
| Clinical Research | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Biotechnology | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Comparative Medicine | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Research Centers in Minority Institutions | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Research Centers | 5 | $11,652 | 6 | $14,471 | 6 | $13,024 | 0 | -$1,447 |
| Other Research: | ||||||||
| Research Careers | 43 | $7,430 | 49 | $7,910 | 48 | $7,515 | -1 | $396 |
| Cancer Education | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Cooperative Clinical Research | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Minority Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Other | 29 | 2,780 | 28 | 2,825 | 27 | 2,543 | -1 | -283 |
| Other Research | 72 | $10,210 | 77 | $10,735 | 75 | $10,057 | -2 | -$678 |
| Total Research Grants | 927 | $363,992 | 935 | $372,138 | 855 | $317,187 | -80 | -$54,951 |
| Ruth L Kirchstein Training Awards: | FTTPs | FTTPs | FTTPs | FTTPs | ||||
| Individual Awards | 130 | $5,894 | 132 | $6,133 | 126 | $5,854 | -6 | -$279 |
| Institutional Awards | 165 | 8,306 | 169 | 8,633 | 161 | 8,224 | -8 | -409 |
| Total Research Training | 295 | $14,200 | 301 | $14,766 | 287 | $14,079 | -14 | $-687 |
| Research & Develop. Contracts | 37 | $18,155 | 41 | $21,500 | 41 | $19,450 | 0 | -$2,050 |
| (SBIR/STTR) (non-add) | (0) | (29) | (0) | (155) | (0) | (133) | (0) | (-22) |
| Intramural Research | 60 | 41,489 | 67 | 42,500 | 67 | 36,492 | 0 | -6,008 |
| Res. Management & Support | 69 | 21,057 | 73 | 23,500 | 73 | 21,150 | 0 | -2,350 |
| Res. Management & Support (SBIR Admin) (non-add) | (0) | (0) | (0) | (0) | (0) | (0) | (0) | (0) |
| Construction | 0 | 0 | 0 | 0 | ||||
| Buildings and Facilities | 0 | 0 | 0 | 0 | ||||
| Total, NIDCD | 129 | $458,893 | 140 | $474,404 | 140 | $408,358 | 0 | -$66,046 |
Major Changes in the Fiscal Year 2020 President’s Budget Request
Major changes by budget mechanism and/or budget activity detail are briefly described below. Note that there may be overlap between budget mechanism and activity detail and these highlights will not sum to the total change for the FY 2020 President’s Budget for NIDCD, which is $66.0 million below FY 2019, for a total of $408.4 million.
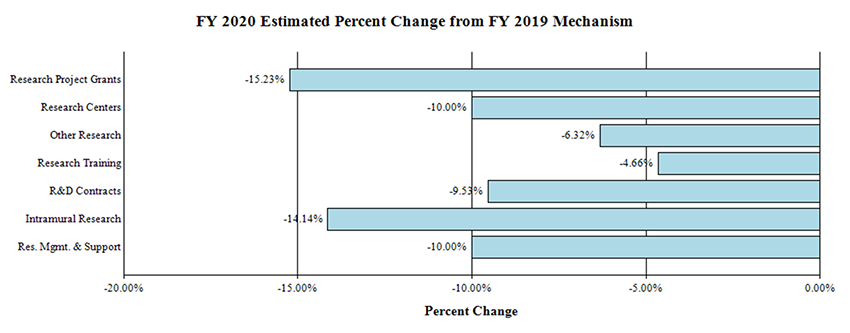
Research Project Grants (RPGs) (-$52.8 million; total $294.1 million):
NIDCD will fund 774 RPG awards in FY 2020, a decrease of 78 awards from the FY 2019 Enacted level. This includes 643 non-competing awards (an increase of 19 awards and a decrease of $8.7 million from the FY 2019 Enacted level); 98 competing RPGs (a decrease of 93 awards and $42.1 million from the FY 2019 Enacted level); and 33 SBIR/STTR awards (a decrease of 4 awards and $2.1 million from the FY 2019 Enacted level). Noncompeting awards will be funded at a reduced level, 9.8 percent below their full committed level. The average cost of Competing RPG’s will decrease by 9.8 percent in FY 2020 versus the FY 2019 Enacted level.
Research Centers (-$1.4 million; total of $13.0 million):
NIDCD will fund 6 Center awards in FY 2020, the same number as the FY 2019 Enacted level.
Other Research (-$0.7 million; total of $10.1 million):
NIDCD will fund 75 Other Research awards in FY 2020, a decrease of 2 from the FY 2019 Enacted level.
Research Training Awards (-$0.7 million; total $14.1 million):
NIDCD will fund 287 Full-Time Training Positions (FTTPs) in FY 2020, a decrease of 14 from the FY 2019 Enacted level.
Research and Development Contracts (-$2.0 million; total $19.4 million):
NIDCD will fund 41 R&D Contracts in FY 2020, the same number as the FY 2019 Enacted level.
Intramural Research (-$6.0 million; total $36.5 million):
In FY 2020, Intramural Research will be reduced by 14.1 percent from the FY 2019 Enacted level.
Research Management & Support (-$2.3 million; total $21.1 million):
In FY 2020, Research Management & Support will be reduced by 10.0 percent from the FY 2019 Enacted level.
| (Dollars in Thousands) | ||||||
|---|---|---|---|---|---|---|
| FY 2019 Enacted | $474,404 | |||||
| FY 2020 President's Budget | $408,358 | |||||
| Net change | -$66,046 | |||||
| CHANGES | FY 2020 President's Budget | Change from FY 2019 Enacted | ||||
| FTEs | Budget Authority | FTEs | Budget Authority | |||
| A. Built-in: | ||||||
| 1. Intramural Research: | ||||||
| a. Annualization of January 2019 pay increase & benefits | $11,831 | $1 | ||||
| b. January FY 2020 pay increase & benefits | 11,831 | 43 | ||||
| c. Paid days adjustment | 11,831 | 45 | ||||
| d. Differences attributable to change in FTE | 11,831 | 0 | ||||
| e. Payment for centrally furnished services | 6,845 | -684 | ||||
| f. Cost of laboratory supplies, materials, other expenses, and non-recurring costs | 17,816 | 0 | ||||
| Subtotal | -$596 | |||||
| 2. Research Management and Support: | ||||||
| a. Annualization of January 2019 pay increase & benefits | $12,512 | $0 | ||||
| b. January FY 2020 pay increase & benefits | 12,512 | 45 | ||||
| c. Paid days adjustment | 12,512 | 47 | ||||
| d. Differences attributable to change in FTE | 12,512 | 0 | ||||
| e. Payment for centrally furnished services | 2,342 | -234 | ||||
| f. Cost of laboratory supplies, materials, other expenses, and non-recurring costs | 6,295 | 0 | ||||
| Subtotal | -$142 | |||||
| Subtotal, Built-in | -$738 | |||||
| CHANGES | FY 2020 President's Budget | Change from FY 2019 Enacted | ||
|---|---|---|---|---|
| No. | Amount | No. | Amount | |
| B. Program: | ||||
| 1. Research Project Grants: | ||||
| a. Nonecompeting | 643 | $244,990 | 19 | -$8,679 |
| b. Competing | 98 | 36,275 | -93 | -42,073 |
| c. SBIR/STTR | 33 | 12,842 | -4 | -2,073 |
| Subtotal, RPGs | 774 | $294,107 | -78 | -$52,825 |
| 2. Research Centers | 6 | $13,024 | 0 | -1,447 |
| 3. Other Research | 75 | 10,057 | -2 | -678 |
| 4. Research Training | 287 | 14,079 | -14 | -687 |
| 5. Research and development contracts | 41 | 19,450 | 0 | -2,050 |
| Subtotal, Extramural | $350,716 | -$57,688 | ||
| FTEs | FTEs | |||
| 6. Intramural Research | 67 | $36,492 | 0 | -$5,412 |
| 7. Research Management and Support | 73 | 21,150 | 0 | -2,208 |
| 8. Construction | 0 | 0 | ||
| 9. Buildings and Facilities | 0 | 0 | ||
| Subtotal, Program | 140 | $408,358 | 0 | -$65,308 |
| Total changes | -$66,046 | |||
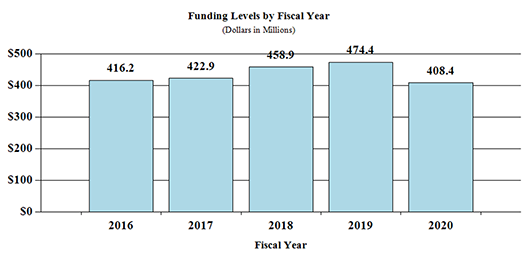
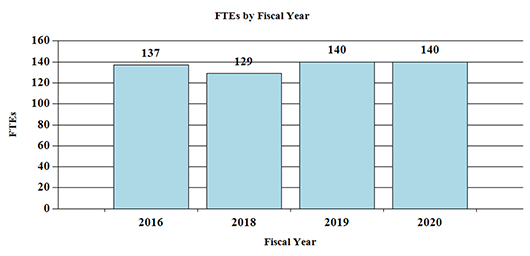
Fiscal Year 2020 Budget Graphs
History of Budget Authority and FTEs:
Distribution by Mechanism:
Change by Selected Mechanism:
| FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY2019 CR | |||||
|---|---|---|---|---|---|---|---|---|
| Extramural Research | FTE | Amount | FTE | Amount | FTE | Amount | FTE | Amount |
| Detail | ||||||||
| Hearing and Balance | $222,600 | $229,371 | $196,972 | -$32,399 | ||||
| Taste and Smell | 65,062 | 67,041 | 57,572 | -9,470 | ||||
| Voice, Speech, and Language | 108,685 | 111,991 | 96,172 | -15,819 | ||||
| Subtotal, Extramural | $396,348 | $408,404 | $350,716 | -$57,688 | ||||
| Intramural Research | 60 | $41,489 | 67 | $42,500 | 67 | $36,492 | 0 | -6,008 |
| Research Management & Support | 69 | $21,057 | 73 | $23,500 | 73 | $21,150 | 0 | -2,350 |
| TOTAL | 129 | $458,893 | 140 | $474,404 | 140 | $408,358 | 0 | -66,046 |
| 1Includes FTEs whose payroll obligations are supported by the NIH Common Fund. | ||||||||
| PHS Act/Other Citation | U.S. Code Citation | 2019 Amount Authorized | FY 2019 Operating Level | 2020 Amount Authorized | FY 2020 President's Budget | |
|---|---|---|---|---|---|---|
| Research and Investigation | Section 301 | 42§241 | Indefinite | $474,404,000 | Indefinite | $408,358,000 |
| National Institute on Deafness and Other Communication Disorders | Section 401(a) | 42§281 | Indefinite | Indefinite |
| Fiscal Year | Budget Estimate to Congress | House Allowance | Senate Allowance | Appropriation |
|---|---|---|---|---|
| 1 Budget Estimate to Congress includes mandatory financing. | ||||
| 2011 | $429,007,000 | $428,331,000 | $418,833,000 | |
| Rescission | $3,677,604 | |||
| 2012 | $426,043,000 | $426,043,000 | $410,482,000 | $417,061,000 |
| Rescission | $788,245 | |||
| 2013 | $417,297,000 | $418,562,000 | $416,272,755 | |
| Rescission | $832,546 | |||
| Sequestration | ($20,894,030) | |||
| 2014 | $422,936,000 | $420,125,000 | $404,049,000 | |
| Rescission | $0 | |||
| 2015 | $403,933,000 | $405,302,000 | ||
| Rescission | $0 | |||
| 2016 | $416,241,000 | $412,366,000 | $424,860,000 | $423,031,000 |
| Rescission | $0 | |||
| 20171 | $422,936,000 | $434,126,000 | $441,778,000 | $436,875,000 |
| Rescission | $0 | |||
| 2018 | $325,846,000 | $443,624,000 | $451,768,000 | $459,974,000 |
| Rescission | $0 | |||
| 2019 | $423,992,000 | $465,467,000 | $474,653,000 | $474,404,000 |
| Rescission | $0 | |||
| 2020 | $408,358,000 | |||
| FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY 2019 | ||||
|---|---|---|---|---|---|---|---|
| BA | $458,893,000 | $474,404,000 | $408,358,000 | -$66,046,000 | |||
| FTE | 129 | 140 | 140 | 0 | |||
Program funds are allocated as follows: Competitive Grants/Cooperative Agreements; Contracts; Direct Federal/Intramural and Other.
Director's Overview
At least 46 million people in the United States have a hearing or other communication disorder.1 For these individuals, the basic components of communication (sensing, interpreting, and responding to people and things in our environment) can be challenging. The National Institute on Deafness and Other Communication Disorders (NIDCD) manages a broad intramural and extramural portfolio of basic and clinical research focused on human communication research and their associated disorders in three program areas: hearing and balance; taste and smell; and voice, speech, and language.
NIDCD Research Advances: Extraordinary research opportunities have led to scientific breakthroughs in the study of genes, proteins, sensory and supporting cells, and molecular processes that directly affect our understanding of communication disorders. In the past year, NIDCD-supported research activities have led to advances in the following areas of communication science:
Exploring the Next Frontier - Treating Hereditary Deafness with Gene Editing2: About 50 to 60 percent of hearing loss at birth is due to genetic factors3, and treatment options for hereditary deafness are limited. Mutations in a particular gene, TMC1, are known to cause hereditary deafness in humans and mice. The mutation causes the death of sensory hair cells in the cochlea of the inner ear. To prevent hair cell death and the resulting progressive hearing loss in mice with the TMC1 mutation, NIDCD-supported scientists used the CRISPR-Cas9 gene-editing system to remove the mutation and disable the gene. The researchers developed a novel approach to deliver the gene-editing complex into the inner ears of newborn mice by packaging the gene-editing complexes in lipids (fats) that form structures called liposomes. The liposome-packaged complexes move readily through cell membranes into cells. As a result, substantially more hair cells survived in the ears of treated compared to untreated mice. This approach also significantly reduced progressive hearing loss. This novel strategy may lead to new therapies for hearing loss caused by inherited genetic mutations.
Building on Basic Science - Prevalence and Risk Factors for Phantom Odor Perception4: A new NIDCD study found that 6.5 percent of Americans over the age of 40 experience phantom odor perception, defined as the sensation of an unpleasant, bad, or burning odor without an identifiable source. Individuals who perceive phantom odors can have a marked reduction in quality of life. This study was the first to use nationally representative data to examine the prevalence of and risk factors for phantom odors in the U.S. Middle-aged women, in particular, were more likely to experience phantom odors compared to older women. People with head trauma, those reporting dry mouth symptoms, and those in poorer health reported phantom odors more frequently.The study could inform future research related to the sensory or neural components of perception of phantom odors.
Transformational Tools and Technologies - Personalized Voices for People with Severely Impaired Speech5: For some individuals with severe speech impairment, communicating is a daily challenge that relies upon use of a computer to generate voice. However, the synthetic voices that are produced are usually a poor representation of a natural human voice and many people must use the same generic version. To address this problem, NIDCD voice scientists have developed a personalized text-to-speech augmentative and alternative communication (AAC) device called VocaliD. This device involves blending the speech of two individuals – a donor and a recipient – to recreate the recipient’s natural voice. A donor voice is matched with a recipient in terms of gender, age, region of origin, and other characteristics to produce a more personalized synthetic voice. By commercializing VocaliD, NIDCD-funded scientists have refined the technology, automating certain steps and making the entire process of creating personalized, synthetic voices faster and more efficient. These improvements will advance speech synthesis while humanizing machine-mediated spoken interaction for AAC devices and beyond.
NIDCD Plans, Priorities, and Challenges for the Future: NIDCD continues its support of new and competing Research Project Grants from investigators who have innovative ideas. NIDCD prioritizes its research investments to identify the most promising opportunities for human communication research, including:
Transformational Tools and Technologies - Accessible and Affordable Hearing Health Care for Adults: Hearing loss is a major public health issue, and yet it remains a neglected part of most mainstream models of health care. Approximately 15 percent of American adults report some degree of hearing loss.6 Despite this, most people do not seek or do not receive hearing health care. For many of these individuals, assistive technologies such as hearing aids could improve their quality of life, yet only about one in four adults over age 20 who could benefit from hearing aids has ever used them. NIDCD prioritizes and continues to support initiatives that promote innovative clinical and translational research to improve accessible and affordable hearing health care, such as identifying barriers to care, assessing novel service delivery and screening models, and multidisciplinary approaches to improve communication and quality of life outcomes for adults with hearing loss.
Exploring the Next Frontier - Dissertation Fellowship and Career Development Awards for Audiologists: NIDCD places great emphasis on supporting training and career development of young and early-stage investigators to ensure an innovative cadre of qualified communication scientists enter the biomedical research workforce. NIDCD supports a Research Dissertation Fellowship for audiologists holding an Au.D. to support comprehensive, rigorous biomedical research training, and dissertation research leading to a Ph.D. in the biomedical, behavioral, or clinical sciences. Further, NIDCD supports two Mentored Career Development Awards for postdoctorate Au.D./Ph.D. audiologists who have the potential to become productive, independent investigators in health-related research fields relevant to NIDCD's mission.
Overall Budget Policy:
The FY 2020 President’s Budget request for NIDCD is $408.4 million, a decrease of $66.0 million or 13.9 percent compared with the FY 2019 Enacted level. Noncompeting RPGs will be awarded at a reduced level, 9.8 percent below their full commited level. The average cost of Competing RPGs will decrease by 9.8 percent in FY 2020 versus the FY 2019 Enacted level. Competing RPGs will achieve a 10 percent success rate. The reduction of 14.1 percent for Intramural Research in FY 2020 equals the aggregate reduction for Extramural funding mechanisms. Research Management & Support will be reduced by 10 percent from the FY 2019 Enacted level.
NIDCD allocates a portion of the funds available for competing RPGs to High Program Priority (HPP) projects outside of the automatic pay line. NIDCD will continue emphasizing faculty, postdoctoral, and student training in our three program areas: hearing and balance; taste and smell; and voice, speech and language sciences. Postdoctoral fellows who show high promise as an emerging independent investigator will be encouraged to launch their careers through the Pathways to Independence (K99-R00) program.
Program Descriptions and Accomplishments
Hearing and Balance Program: Loss of hearing or balance imposes a significant social and economic burden upon individuals, their families, and the communities in which they live. Hearing and balance disorders also decrease quality of life and cross all ethnic and socioeconomic lines. Among adults, hearing loss is the third most common chronic condition in the U.S.7 More than 1 in 20 American children have a dizziness or balance problem.8 In addition, between one and two out of every 1,000 children in the U.S. are born with a detectable level of hearing loss in one or both ears that can affect their speech, language, social, and cognitive development.9 Accordingly, research projects within the NIDCD Hearing and Balance Program encompass over half of NIDCD’s portfolio. NIDCD supports research that will lead to improved treatments for, and prevention of, hearing and balance disorders.
Loud noises can cause permanent hearing loss through damage to the sensitive structures of the auditory system. Researchers have focused on the death of sensory hair cells in the inner ear as the major cause of this damage, but recent animal studies have demonstrated that there is permanent damage by an additional mechanism called cochlear synaptopathy—the extensive, permanent loss of connections between hair cells and the auditory nerve fibers that send sound information to the brain. In 2015, NIDCD held a workshop on cochlear synaptopathy,10 and scientific experts there identified urgent research needs to determine whether or not noise-induced cochlear synaptopathy occurs in humans and if so, how to detect this type of auditory system damage in the clinic.
Guided by the recommendation from this workshop, significant advances have been made toward characterizing the physiology, potential symptoms, and non-invasive behavioral and physiological diagnosis of synaptopathy. For example, one group found evidence for synaptopathy in humans using autopsy tissue. They found that the loss of synaptic connections was much more common than loss of hair cells, even in younger individuals.11 In another study, scientists investigated whether a muscle reflex in the middle ear could be used as a way to detect cochlear synaptopathy, potentially opening the possibility to use this as an early diagnostic test. Researchers found that individuals with tinnitus (ringing in the ears that can be an early sign of hearing loss), who performed normally on hearing tests, had a reduced muscle reflex.12 These studies suggest that cochlear synaptopathy may occur in humans, and researchers may soon be able to develop clinical tests to detect this hearing impairment which is missed by typical hearing tests.
Age-related hearing loss, also known as presbycusis, is the gradual loss of hearing that occurs in most people as they grow older. Approximately two-thirds of Americans 70 years or older currently have hearing loss, and this number is expected to rise significantly as the older adult population grows.13 Hearing loss affects quality of life by impairing communication and by increasing the risk of social isolation. There is also emerging evidence that suggests a relationship between presbycusis and cognitive decline.14 Recent NIDCD-supported research explores the high cost of hearing loss on medical care in the United States. Health care costs were 20 percent higher for those with hearing loss,15 and this trend was even more significant when individuals with hearing loss didn’t have access to hearing health services, like hearing aids, through insurance. Delaying treatment for hearing loss may make it harder for the brain to process sounds over time because of a lack of stimulation of hearing areas in the brain. One recent NIDCD-supported study found that people with hearing loss who used hearing aids had better hearing abilities over time compared to people with hearing loss who did not use hearing aids.16 Another NIDCD-supported study found that people who used hearing aids had enhanced working memory and brain activity patterns, compared with individuals with untreated hearing loss.17
Ménière’s disease is a disorder of the inner ear that causes severe dizziness (vertigo), tinnitus, hearing loss, and a feeling of fullness or congestion in the ear. Some people with Ménière’s disease have vertigo so extreme that they lose their balance and fall. Ménière’s disease can develop at any age, but it is more likely to happen to adults between 40 and 60 years old. The symptoms of Ménière’s disease are associated with the buildup of fluid, endolymph, in the compartments of the inner ear, called the labyrinth, which contains the organs of balance. In Ménière’s disease, the endolymph buildup in the labyrinth may interfere with the normal balance and hearing signals between the inner ear and the brain. NIDCD supported-scientists are determining the mechanisms that control endolymph pressure in the inner ear as a way to better understand the cause of Ménière’s disease and are developing an implantable vestibular prosthesis that can restore balance in severe cases. Further, NIDCD is continuing its long-standing support of the National Temporal Bone Registry and the National Human Ear Tissue Laboratory Resource for Hearing and Balance Research. Through these resources, researchers have access to human inner and middle ear tissues to investigate directly differences between normal and disordered tissue or to validate if findings from animal studies of Ménière’s disease translate into humans.
Individuals with inner ear diseases who visit the emergency room with symptoms of vertigo or dizziness are frequently misdiagnosed, resulting in incorrect treatments. Better diagnostic tools would improve clinical care for individuals with inner ear balance disorders and for those with more dangerous diseases causing vertigo or dizziness, such as stroke. NIDCD-supported scientists are conducting the Acute Video-oculography for Vertigo in Emergency Rooms for Rapid Triage (AVERT) clinical trial to help diagnose vertigo, dizziness, and other balance problems. The team of researchers is using a diagnostic medical device (video-oculography or VOG) to triage individuals who go to emergency room with complaints of vertigo and/or dizziness. This new, non-invasive device measures abnormal eye movements and can assist physicians in accurately diagnosing individuals with vertigo or dizziness, leading to better outcomes at lower cost.
PROGRAM PORTRAIT: Transformational Tools and Technologies - Open Speech Signal Processing
Today’s hearing aids are limited by size and power, which constrains their ability to reduce background noises, a major complaint for individuals with hearing loss. Continued advancements in computer chips should enable new speech enhancement and noise reduction features in the future. Research efforts must be started today so that results describing user benefits will be available to help shape novel concepts into proven algorithms for commercial use. To this end, NIDCD held a workshop in 2014 to discuss the potential value of a research tool that employs open design principles and encourages continued increases in the computing power available in a portable device that runs hearing aid software. Scientific experts at the workshop also discussed ways to accelerate research and to develop bold, new algorithms for the field of acoustic signal processing, speech enhancement, and noise reduction. This will accelerate the pace of research and commercialization for novel acoustic signal processing features for use in portable hearing devices.
Open design engineering practices allow rapid implementation of new features driven by the needs of a single researcher or a large community of users. Research studies performed with open design platforms allow study results to be published and replicated with greater clarity since the exact specifications for the hardware and software are referenced in the study. System enhancements can be shared widely and the benefits from collaborative design and validation procedures acknowledge contributions from all team members -- innovators and users alike.
Following the 2014 workshop, NIDCD developed two Funding Opportunity Announcements (FOAs) on “Open Design Tools for Speech Signal Processing” to develop and support the use of portable acoustic signal processing tools that provide substantial amounts of computing power and reconfigurable real-time speech enhancement/noise reduction software. Research tools developed under these initiatives provide open source hardware and software to support rapid dissemination, reconfiguration, and enhancement of the tools. To date, these FOAs support three academic research projects and three small business innovation research and development projects. Awardees are encouraged to place an emphasis on outreach and dissemination activities to ensure widespread and effective use of these research tools by outside laboratories. Curated websites are online with user support forums, answers to common user questions, and software for download, to engage new users and obtain continued feedback from current users. Agile software development methods are encouraged in order to gain feedback from users, build consensus among users, and identify emerging needs based on ongoing research. The research community can use these tools to lower barriers for hardware and software refinement, accelerate studies with novel acoustic processing algorithms, and facilitate translation of these advances into widespread use in hearing aids, cochlear implants, and consumer electronics devices.
Budget Policy:
The FY 2020 President’s Budget request for the Hearing and Balance program is $197.0 million, a decrease of $32.4 million or 14.1 percent compared with the FY 2019 Enacted level. In FY 2020, the program will continue emphasizing faculty, postdoctoral, and student training on hearing and balance sciences. Postdoctoral fellows who show high promise as emerging independent investigators will be encouraged to launch their careers through the Pathways to Independence (K99-R00) program. Making R01 awards to first-time and early-stage investigators in the area of hearing and balance sciences will be given funding priority.
Taste and Smell Program: Many taste and smell disorders go undiagnosed or unreported. NIDCD supports studies of the chemical senses known as taste, smell, and chemesthesis (chemically provoked irritation) to enhance our understanding of how individuals communicate with their environment and how human chemosensory disorders can be diagnosed and treated. With age, the olfactory (relating to sense of smell) system is less able to regenerate, and our sense of smell declines. Although prevalence estimates vary, it is likely that more than one third of adults over the age of 70 have olfactory deficits.18 Since both taste and smell contribute to flavor perception, such olfactory deficits affect the flavor of foods and consequently food intake, diet, overall nutrition, and health status. NIDCD encourages further studies of age-related decline in olfactory sensitivity including the development of better diagnostic tests to assess chemosensory loss and animal models to study why this decline occurs and how to prevent it.
NIDCD-supported scientists are working to identify treatments for human taste and smell disorders. Humans who lose hair-like structures called cilia within their olfactory system may also lose their ability to detect odors. Working in a mouse model of human olfactory cilia diseases (called ciliopathies), NIDCD-supported scientists used gene therapy to re-express a critical odor-detecting protein, and the mice were once again able to detect odors.19 If this result can be repeated in humans with ciliopathies, it could be used to treat a group of inherited human diseases that can cause loss of smell.
NIDCD is also supporting a clinical trial to try to change how humans perceive sweet taste and our preference for sugar in our diets.A diet rich in sugar intake is known to cause poor health outcomes. The natural human tendency to seek out sweet tastes is challenging, and this trial is modeled on previous work on salt taste, which showed that consuming a low salt diet gradually led people to prefer less salt in their foods. The current trial will take a similar approach, in which people will eat less dietary sugar, and the investigators will determine whether participants gradually prefer less sugar in their diets over the course of a three-month period. If this study establishes that reduced sugar consumption is a valid way to reduce preference for sugar in our diets, it may go to inform policy recommendations concerning reduction of simple sugars in manufactured and prepared foods to provide a healthier diet for everyone.
Taste cells on the tongue respond to chemicals released from food and drink via protein receptors and channels specific to certain taste molecules. To detect sourness in food, specialized channels let protons (Hydrogen ions) released from acidic sour-tasting foods enter proton-sensitive, “sour” taste cells on the tongue. The identity of this “sour detector” protein has been elusive. Now, NIDCD-supported scientists have located a protein called OTOP1 and determined that it forms a channel that allows protons to enter taste cells on the tongue.20 Studies like this one increase our understanding about how humans taste and may help scientists learn to restore a sense of taste to those who have lost it due to disease or injury.
This study also elucidates more than just how humans detect sour taste. For example, OTOP1 is required for the vestibular (balance) system in the inner ear to detect gravity and motion, so it is important for helping maintain balance. Because OTOP1 is also expressed in many other body tissues, including fat, heart, uterus, breast, and the nervous system, these findings can inform understanding of cell signaling processes in other tissues as well. When protons enter cells, they change the acid/base (pH) concentration. Thus, a better understanding of OTOP1 function could also help researchers understand other body processes that involve changes in pH, such as pain sensation, fat metabolism, and pH changes seen in cancer cells.
Budget Policy:
The FY 2020 President’s Budget request for the Taste and Smell program is $57.6 million, a decrease of $9.5 million or 14.1 percent compared with the FY 2019 Enacted level. In FY 2020, the program will continue emphasizing faculty, postdoctoral, and student training on smell and taste sciences. Postdoctoral fellows who show high promise as emerging independent investigators will be encouraged to launch their careers through the Pathways to Independence (K99-R00) program. Making R01 awards to first-time and early-stage investigators in the area of smell and taste sciences will be given funding priority.
Voice, Speech, and Language Program: Disorders involving voice, speech, or language can have an overwhelming effect on an individual’s health and quality of life. These disorders affect people of all ages with or without hearing impairment, including children with autism, those who stutter, and adults with acquired communication disorders. Voice, speech, and language disorders also come at a significant cost. The societal burden of laryngeal disorders is estimated at $11 billion annually due to work-related disability, lost productivity, and direct health care cost.21 Further, nearly eight percent of children ages 3-17 years have had a communication disorder during the past 12 months, according to data from the National Health Interview Survey, 2012.22 By the first grade, roughly five percent of children have noticeable speech disorders.23 In children, delayed speech and language acquisition or impairment are very often significant predictors of future academic, social, vocational, and adaptive outcomes.24
NIDCD supports research to understand diseases and stresses that can harm the voice and clinical research to test new ways to diagnose, treat, or cure voice disorders. Regenerative medicine approaches to treat voice disorders that are caused by damage to the laryngeal system, which includes the vocal folds, focus on engineering replacement tissues and methods to regenerate nerve connections to repair damaged tissue. For example, in one NIDCD-funded study researchers aim to develop injectable biomaterials that will alter the immune response in order to decrease inflammation and improve wound healing for vocal fold scarring. Another study evaluates a new method to treat laryngeal paralysis with an implantable, electric pulse generator to encourage nerve regrowth to the muscles that control voice and swallowing.
Further, significant advances have been made in replacing, engineering, and regenerating laryngeal tissue through the use of stem cell technologies. For example, in two separate studies, NIDCD-funded researchers used stem cells to engineer laryngeal cartilage25 and used progenitor cells (like stem cells, they can differentiate into specific “target” cells) to engineer laryngeal muscle.26 The scientists were then successful in using these generated tissues to repair and reconstruct functional laryngeal cartilage and muscle in an animal model. In another study funded in part by NIDCD, researchers developed a new method that introduced cell-guidance cues at specific times to human mesenchymal stem cells to establish functional tissue constructs. This new technique complements existing methods that require specialized equipment to help guide stem cells to become differentiated tissue.27 These findings help pave the way for scientists to restore laryngeal structure and function and to develop improved treatment options for voice disorders through regenerative medicine.
NIDCD supports a language program which includes research on American Sign Language. Funded research includes studies to elucidate sign language communication systems, their acquisition and development, and their use when spoken language systems are damaged by trauma or degenerative diseases, or when speech is difficult to acquire due to early hearing loss or injury to the nervous system. The NIDCD 2017-2021 Strategic Plan identifies sign language research as a high priority area and outlines objectives to: 1) investigate the acquisition, processing, and neural underpinnings of languages within the visual-manual modality; 2) to identify central and peripheral factors associated with the successful comprehension and use of written language for people who use sign language as their primary way of communication; and 3) to develop methods that promote the acquisition of literacy skills during childhood and improve the reading and writing abilities of people who are deaf and native American Sign Language users.
Studying sign language can also help scientists understand the neurobiology of how humans develop language. In a new study, researchers reported that building of complex phrases, whether signed or spoken, engaged the same brain areas and with similar neural timing.28 Overall, this research enhances understanding of the neurobiology of signed language, which could provide a translational foundation for treating injury to the language system, for employing signs or gestures in therapy for children or adults, and for diagnosing language impairment in individuals who are deaf. Further, NIDCD is funding research on sign languages from foreign countries because those sign languages can be developed among a small group of people with little to no outside influence and no direct linguistic input. Emerging sign languages can be used to model the essential elements and organization of natural language and to inform researchers about the complex interplay between natural human language abilities, language environment, and language learning outcomes.
NIDCD has partnered with the National Institute on Aging and the NIH Office of the Director to grow the community of human communication scientists actively engaged on two trans-NIH initiatives, respectively – the Alzheimer’s Disease and Alzheimer’s Disease-Related Dementias (AD/ADRD) Initiative and the Investigation of Co-Occurring Conditions Across the Lifespan to Understand Down Syndrome (INCLUDE) Initiative. To date, fifteen NIDCD grantees have received supplemental funding. These initiatives expand opportunities for NIDCD-funded scientists to identify new implications for their research. For example, two studies focus on adapting assistive communication technologies to help extend cognitive, written, and spoken abilities in individuals with AD/ADRD. NIDCD researchers will expand neurobehavioral assessment studies to identify which children with Down syndrome (DS) are also at increased risk for having autism. Another project aims to identify lifespan patterns of speech intelligibility in individuals with DS from childhood into adulthood. These research areas were identified by advocates and public stakeholders as being particularly relevant to improving the quality of life for individuals with voice, speech, and language disorders, AD/ADRD, and DS.
Budget Policy:
The FY 2020 President’s Budget request for the Voice, Speech, and Language program is $96.2 million, a decrease of $15.8 million or 14.1 percent compared with the FY 2019 Enacted level. In FY 2020, the program will continue emphasizing faculty, postdoctoral, and student training on voice, speech, and language sciences. Postdoctoral fellows who show high promise as emerging independent investigators will be encouraged to launch their careers through the Pathways to Independence (K99-R00) program. Making R01 awards to first-time and early-stage investigators in the area of voice, speech, and language sciences will be given funding priority.
Intramural Research Program (IRP): The NIDCD Intramural Research Program conducts basic and clinical research in human communication, with a primary focus on hearing and balance.
NIDCD intramural researchers have found a new way to explain the hearing loss caused by cisplatin, a powerful drug used to treat many forms of cancer. Cisplatin and other common and effective platinum-based chemotherapy drugs cause hearing loss through ototoxicity—damage to the delicate hair cells in the inner ear.29 As hearing loss is often associated with conditions such as isolation and depression, helping to preserve hearing in individuals who use these life-saving chemotherapy drugs would greatly contribute to maintaining the quality of their lives. In previous studies, researchers focused on why the inner ear is more vulnerable to cisplatin ototoxicity than other areas in the body. The NIDCD research team, however, found that in mice and humans, cisplatin remains in the inner ear long after it is already eliminated from other areas of the body. The research team saw the highest accumulation of cisplatin in a part of the inner ear called the stria vascularis, which is responsible for maintaining the positive electrical charge in inner ear fluid that certain cells need to detect sound. This research suggests that if we can find ways to avoid cisplatin from entering and building up in the stria vascularis, we might be able to prevent hearing loss and maintain quality of life for cancer survivors.
In another study, NIDCD intramural researchers, in collaboration with other NIDCD-funded researchers, found a small-molecule drug that preserved hearing in a mouse model of an inherited form of progressive human deafness (DFNA27).30 Researchers were able to specifically identify the location in the genome responsible for this form of deafness, which was previously elusive. By following these genetic leads, researchers found a novel and unexpected cellular pathway involved in this form of deafness. They used what was known about this pathway to find and use a small molecule drug therapy that could partially counteract this form of deafness in mice. This drug partially restored hearing, especially at lower frequencies, and saved some sensory hair cells. If additional studies show that small-molecule-based drugs are effective in treating DFNA27 deafness in people, it’s possible that using similar, targeted approaches might work for other inherited forms of progressive hearing loss.
PROGRAM PORTRAIT: Exploring the Next Frontier – Otolaryngology Surgeon-Scientist Program
Bringing the enormous opportunities and potential of the recent advances in genetics, molecular biology, and cellular biology to the benefit of human communication disorders requires otolaryngology surgeon-scientists who can conceive, develop, and test novel diagnostic and therapeutic strategies through translational research that bridges basic science, medicine, and surgery. The NIDCD Otolaryngology Surgeon-Scientist Program (OSSP) advances NIDCD’s programmatic priority to promote biomedical and clinical research training and career development. This translational and clinical research program complements the rich basic science research within NIDCD and other institutes in the NIH intramural program. Areas of priority and opportunity include disorders affecting hearing, balance, taste, smell, voice, speech, and language. Current programs have advanced the capability for preclinical and clinical studies for interventions in these disorders.
The OSSP has developed partnerships with academic and government hospitals, which include Johns Hopkins University Medical Center, Georgetown University Medical Center, George Washington University Medical Center, and Walter Reed National Military Medical Center. Joint educational, training, and clinical opportunities and appointments with these institutions further increase the depth and breadth of training in the OSSP. The OSSP offers flexibility to customize the site(s), type, and extent of clinical and surgical activity to best meet the career development objectives of the surgeon-scientist.
NIDCD has been training otolaryngologist-scientists since 1995, with leading NIH and academic otolaryngologist surgeon-scientists among its alumni. The program is a junior faculty level career development program that is comparable to extramural mentored K-award programs. Most participants have already completed an otolaryngology residency or subspecialty fellowship. Typical sub-specialty fellowship training often does not provide adequate research experience upon which to build an enduring and robust independent research career, particularly when clinical research with human volunteers is involved. The purpose of the OSSP is to provide the necessary training—with formal research and career mentorship and strong support—for a successful transition to an independent tenure-track surgeon-scientist position.
NIDCD surgeon-scientists lead significant research advances through the following programs:
- Scientists in the Translational Tumor Immunology Program are using various therapeutic approaches to reverse local immunosuppression within the tumor microenvironment to enhance responses to immune-activating anti-cancer treatments against head and neck squamous cell carcinoma, the most common form of head and neck cancer. Several clinical trials aimed at improving response to therapy and survival in patients with head and neck cancer are being developed.
- The Inner Ear Gene Therapy Program is focused on developing gene therapy as a treatment option for hearing loss and dizziness. The laboratory is developing ways to delivery gene therapy to the mammalian inner ear in vivo and is currently testing whether the delivery of normal copies of the mutated genes into animal models of hereditary hearing loss can restore hearing and balance. In addition, the laboratory is studying whether genome editing can be used as a form of gene therapy to restore hearing and balance in animal models.
- Researchers in the Auditory Development and Restoration Program are investigating the development and function of adult cochlear cell types and the pathophysiological processes that result in their structural and functional degeneration with the long-term goal of establishing a basis for hearing restoration in the adult inner ear. Investigations may provide insight into hearing loss related to pathology in the cochlear lateral wall, as well as potential avenues for therapeutic targeting of this pathology.
- The Integrative Therapeutics Program is focused on optimizing the use of cisplatin chemotherapy for treatment of head and neck squamous cell carcinoma (HNSCC). A clinical trial studying hearing loss and the effects of statins (drugs to lower cholesterol) in people with HNSCC treated with cisplatin is underway. Cisplatin is the most commonly used drug for HNSCC, but it can cause hearing loss.
Budget Policy:
The FY 2020 President’s Budget request for the Intramural Research program is $36.5 million, a decrease of $6.0 million or 14.1 percent compared with the FY 2019 Enacted level.The NIDCD Intramural Research Program has been a leader in research on hereditary hearing impairment and has identified many genes whose mutation causes hearing loss. In FY 2020, NIDCD intramural scientists will continue their efforts in identifying mutation in genes, which impacts hearing, and identifying or studying additional genes involved in communication disorders in humans and animal models.
Research Management and Support (RMS) Program: NIDCD RMS activities provide administrative, budgetary, logistical, and scientific support in the review, award, and monitoring of research grants, training awards, and research and development contracts. RMS functions also include strategic planning, coordination, and evaluation of the Institute’s programs, regulatory compliance, international coordination, and liaison with other Federal agencies, Congress, and the public. The Institute currently supports approximately 1,400 research grants, training awards, and R&D contracts.
In 2018, NIDCD celebrated 30 years of supporting human communications research and research training, since the Institute was established by law on October 28, 1988. NIDCD marked the occasion by developing an NIDCD anniversary web page to include information about NIDCD’s mission and history, and stories based on interviews with the individuals who worked together to establish NIDCD.
Also, 2018 marks the 10th anniversary of the NIDCD public education campaign - It’s a Noisy Planet. Protect Their Hearing.® The campaign’s purpose is to prevent noise-induced hearing loss (NIHL)—hearing loss caused by exposure to loud noise—by increasing awareness of NIHL among children ages 8 to 12, parents, and educators. The Noisy Planet campaign has reached approximately six million people. As it moves into its second decade, the campaign will continue to spread the NIHL prevention message in English and Spanish to children, parents, school nurses, teachers, audiologists, and others.
Budget Policy:
The FY 2020 President’s Budget request for the Research Management and Support program is $21.2 million, a decrease of $2.4 million or 10.0 percent compared with the FY 2019 Enacted level.
| FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY2019 | |
|---|---|---|---|
| OBJECT CLASSES | FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY2019 |
| 1Includes FTEs whose payroll obligations are supported by the NIH Common Fund. | |||
| Total compensable work years | |||
| Full-time equivalent | 140 | 140 | 0 |
| Full-time equivalent of overtime and holiday hours | 0 | 0 | 0 |
| Average ES salary | $190 | $190 | $0 |
| Average GM/GS grade | $12.4 | $12.4 | 0.0 |
| Average GM/GS salary | $115 | $115 | $0 |
| Average salary, grade established by act of July 1, 1944 (42 U.S.C. 207) | $115 | $115 | $0 |
| Average salary of ungraded positions | $102 | $102 | $0 |
| Personnel Compensation | |||
| 11.1 Full-Time Permanent | 11,694 | 11,739 | 44 |
| 11.3 Other Than Full-Time Permanent | 4,671 | 4,689 | 18 |
| 11.5 Other Personnel Compensation | 396 | 398 | 2 |
| 11.7 Military Personnel | 127 | 132 | 4 |
| 11.8 Special Personnel Services Payment | 1,702 | 1,708 | 6 |
| 11.9 Subtotal Compensation | $18,591 | $18,665 | $74 |
| 12.1 Civilian Personnel Benefits | 5,514 | 5,618 | 104 |
| 12.2 Military Personnel Benefits | 59 | 61 | 2 |
| 13.0 Benefits to Former Personnel | 0 | 0 | 0 |
| Subtotal Pay Costs | $24,163 | $24,344 | $181 |
| 21.0 Travel & Transportation of Persons | 338 | 304 | -34 |
| 22.0 Transportation of Things | 46 | 37 | -9 |
| 23.1 Rental Payments to GSA | 0 | 0 | 0 |
| 23.2 Rental Payments to others | 0 | 0 | 0 |
| 23.3 Communication, Utilities & Misc. Charges | 125 | 125 | 0 |
| 24.0 Printing & Reproduction | 0 | 0 | 0 |
| 25.1 Consulting Services | 142 | 97 | -45 |
| 25.2 Other Services | 6,819 | 4,638 | -2,181 |
| 25.3 Purchase of goods and services from government account | 45,330 | 39,480 | -5,850 |
| 25.4 Operation & Maintenance of Facilities | 30 | 30 | 0 |
| 25.5 R&D Contracts | 4,268 | 3,348 | -920 |
| 25.6 Medical Care | 182 | 182 | 0 |
| 25.7 Operation & Maintenance of Equipment | 1,496 | 1,496 | 0 |
| 25.8 Subsistence & Support of Persons | 32 | 32 | 0 |
| 25.0 Subtotal Other Contractual Services | $58,300 | $49,304 | -$8,996 |
| 26.0 Supplies & Materials | 1,821 | 1,321 | -500 |
| 31.0 Equipment | 2,707 | 1,657 | -1,050 |
| 32.0 Land and Structures | 0 | 0 | 0 |
| 33.0 Investment & Loans | 0 | 0 | 0 |
| 41.0 Grants, Subsidies & Contributions | 386,904 | 331,266 | -55,638 |
| 42.0 Insurance Claims & Indemnities | 0 | 0 | 0 |
| 43.0 Interest & Dividends | 0 | 0 | 0 |
| 44.0 Refunds | 0 | 0 | 0 |
| Subtotal Non-Pay Costs | $450,241 | $384,014 | -$66,227 |
| Total Budget Authority by Object Class | $474,404 | $408,358 | -$66,046 |
| OBJECT CLASSES | FY 2019 Enacted | FY 2020 President's Budget | FY 2020 +/- FY2019 |
|---|---|---|---|
| Personnel Compensation | |||
| Full-Time Permanent (11.1) | $11,694 | $11,739 | $44 |
| Other Than Full-Time Permanent (11.3) | 4,671 | 4,689 | 18 |
| Other Personnel Compensation (11.5) | 396 | 398 | 2 |
| Military Personnel (11.7) | 127 | 132 | 4 |
| Special Personnel Services Payments (11.8) | 1,702 | 1,708 | 6 |
| Subtotal Personnel Compensation (11.9) | $18,591 | $18,665 | $74 |
| Civilian Personnel Benefits (12.1) | $5,514 | $5,618 | $104 |
| Military Civilian Personnel Benefits (12.2) | 59 | 61 | 2 |
| Benefits to Former Personnel (13.0) | 0 | 0 | 0 |
| Subtotal Pay Costs | $24,163 | $24,344 | $181 |
| Travel & Transportation of Persons (21.0) | $338 | $304 | -$34 |
| Transportation of Things (22.0) | 46 | 37 | -9 |
| Rental Payments to Others (23.2) | 0 | 0 | 0 |
| Communications, Utilities & Misc. Charges (23.3) | 125 | 125 | 0 |
| Printing & Reproduction (24.0) | 0 | 0 | 0 |
| Other Contractual Services | |||
| Consultant Services (25.1) | 142 | 97 | -45 |
| Other Services (25.2) | 6,819 | 4,638 | -2,181 |
| Purchases from government accounts (25.3) | 30,418 | 25,516 | -4,902 |
| Operation & Maintenance of Facilities (25.4) | 30 | 30 | 0 |
| Operation & Maintenance of Equipment (25.7) | 1,496 | 1,496 | 0 |
| Subsistence & Support of Persons (25.8) | 32 | 32 | 0 |
| Subtotal Other Contractual Services | $38,938 | $31,809 | -$7,129 |
| Supplies and Materials (26.0) | $1,821 | $1,321 | -$500 |
| Subtotal Non-Pay Costs | $41,268 | $33,596 | -$7,671 |
| Total Administrative Costs | $65,431 | $57,940 | -$7,491 |
| OFFICE/DIVISION | FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Civilian | Military | Total | Civilian | Military | Total | Civilian | Military | Total | |
| Fiscal Year | Average GS Grade | ||||||||
| Division of Extramural Activities | |||||||||
| Direct: | 19 | - | 19 | 19 | - | 19 | 19 | - | 19 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 19 | - | 19 | 19 | - | 19 | 19 | - | 19 |
| Division of Intramural Research Program | |||||||||
| Total: | 2 | - | 2 | 3 | - | 3 | 3 | - | 3 |
| Total: | 128 | 1 | 129 | 139 | 1 | 140 | 139 | 1 | 140 |
| Includes FTEs whose payroll obligations are supported by the NIH Common Fund. | |||||||||
| FTEs supported by funds from Cooperative Research and Development Agreements. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2018 | 12.4 | ||||||||
| 2019 | 12.4 | ||||||||
| 2020 | 12.4 | ||||||||
| GRADE | FY 2018 Final | FY 2019 Enacted | FY 2020 President's Budget |
|---|---|---|---|
| 1Includes FTEs whose payroll obligations are supported by the NIH Common Fund. | |||
| Total, ES Positions | 1 | 1 | 1 |
| Total, ES Salary | 189,600 | 189,600 | 189,600 |
| GM/GS-14 | 16 | 19 | 19 |
| GM/GS-13 | 21 | 23 | 23 |
| GS-12 | 22 | 24 | 24 |
| GS-11 | 4 | 4 | 4 |
| GS-10 | 0 | 0 | 0 |
| GS-9 | 9 | 9 | 9 |
| GS-8 | 5 | 5 | 5 |
| GS-7 | 0 | 0 | 0 |
| GS-6 | 0 | 0 | 0 |
| GS-5 | 0 | 0 | 0 |
| GS-4 | 2 | 2 | 2 |
| GS-3 | 1 | 1 | 1 |
| GS-2 | 1 | 1 | 1 |
| GS-1 | 0 | 0 | 0 |
| Subtotal | 104 | 112 | 112 |
| Assistant Surgeon General | 0 | 0 | 0 |
| Director Grade | 0 | 0 | 0 |
| Senior Grade | 1 | 1 | 1 |
| Full Grade | 0 | 0 | 0 |
| Senior Assistant Grade | 0 | 0 | 0 |
| Assistant Grade | 0 | 0 | 0 |
| Subtotal | 1 | 1 | 1 |
| Total permanent positions | 100 | 111 | 111 |
| Total positions, end of year | 148 | 159 | 159 |
| Total full-time equivalent (FTE) employment, end of year | 129 | 140 | 140 |
| Average GM/GS grade | 12.4 | 12.4 | 12.4 |
| Average GM/GS salary | 114,582 | 114,582 | 114,582 |
- www.nidcd.nih.gov/about/strategic-plan/2017-2021
- www.ncbi.nlm.nih.gov/pubmed/29258297
- www.cdc.gov/ncbddd/hearingloss/data.html
- jamanetwork.com/journals/jamaotolaryngology/fullarticle/2696525
- projectreporter.nih.gov/project_info_description.cfm?aid=9509403
- www.cdc.gov/nchs/data/series/sr_10/sr10_260.pdf
- www.cdc.gov/nchs/data/series/sr_10/sr10_260.pdf
- www.nidcd.nih.gov/news/2016/more-1-20-us-children-have-dizziness-and-balance-problems
- onlinelibrary.wiley.com/doi/10.1111/ppe.12499
- www.nidcd.nih.gov/research/workshops/synaptopathy-and-noise-induced-hearing-loss-animal-studies-and-implications/2015/
- www.ncbi.nlm.nih.gov/pubmed/30099118
- www.ncbi.nlm.nih.gov/pubmed/?term=29181442
- jamanetwork.com/journals/jamaotolaryngology/fullarticle/2606784
- www.ncbi.nlm.nih.gov/pubmed/30248173
- www.ncbi.nlm.nih.gov/pubmed/?term=29797584
- www.ncbi.nlm.nih.gov/pubmed/?term=29677689
- www.ncbi.nlm.nih.gov/pubmed/?term=29729278
- NIDCD Epidemiology and Statistics Branch calculation
- www.ncbi.nlm.nih.gov/pubmed/30061191
- www.ncbi.nlm.nih.gov/pubmed/29371428
- www.ncbi.nlm.nih.gov/pubmed/22549455, www.ncbi.nlm.nih.gov/pubmed/22544473
- https://www.cdc.gov/nchs/products/databriefs/db205.htm
- www.ahrq.gov/downloads/pub/prevent/pdfser/speechsyn.pdf
- www.ncbi.nlm.nih.gov/pubmed/18695010, www.ncbi.nlm.nih.gov/pubmed/17883441, www.ncbi.nlm.nih.gov/pubmed/15679523
- www.ncbi.nlm.nih.gov/pubmed/29238978
- www.ncbi.nlm.nih.gov/pubmed/?term=28842993
- www.ncbi.nlm.nih.gov/pmc/articles/PMC6009435/
- www.ncbi.nlm.nih.gov/pubmed/29615785
- www.nature.com/articles/s41467-017-01837-1
- www.cell.com/cell/fulltext/S0092-8674(18)30729-3
* Note: PDF files require a viewer such as the free Adobe Reader.

