Chief, Section on Sensory Cell Development and Function

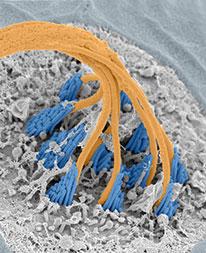
Dr. Kindt received a B.S. in molecular biology and biochemistry from the University Wisconsin-Eau Claire, and a Ph.D. in biomedical sciences from the University of California, San Diego, where she studied the function and development of mechanosensory circuits in Caenorhabditis elegans in the laboratory of William Schafer. During a postdoctoral fellowship with Teresa Nicolson at the Vollum Institute, she used a combination of scanning electron microscopy and in vivo calcium imaging to investigate the role of the primary cilium in developing hair cells (figure 1). Dr. Kindt joined the NIDCD as an investigator in 2013. Her laboratory, the Section on Sensory Cell Development and Function, uses molecular and microscopy-based methods to examine sensory cell function and development in the zebrafish model system.
Laboratory of Cellular Biology
Genetics and neuroimaging of hair cell development in zebrafish

